Introduction
In laboratory environments, accuracy and precision are non-negotiable. While much attention is given to equipment and chemicals, one essential resource is often underestimated: water. The quality of water used in experiments directly impacts the reliability of results, the longevity of instruments, and the integrity of research. Trace impurities, such as ions and microorganisms, can alter outcomes, cause contamination, or damage sensitive equipment. That’s why every laboratory—regardless of field—requires a dependable deionized (DI) water system. This article explores what DI water is, how it’s produced, where it’s used, and why a reliable DI water system is essential for any laboratory setting.

What is Deionized Water?
Deionized water, also known as demineralized water, is water that has had nearly all dissolved mineral ions removed—both cations like calcium, sodium, and iron, and anions like chloride and sulfate. The result is chemically neutral water with high purity that won’t interfere with chemical reactions or analytical procedures.
Key Characteristics of DI Water
High Purity – Minimal dissolved solids or contaminants
Low Conductivity – Poor conductor of electricity due to the absence of ions
Neutral pH – Typically near 7, although exposure to air can shift pH
Corrosive Nature – Can be aggressive to certain metals (e.g., copper, aluminum) due to lack of buffering ions
Flat Taste – Lacks minerals found in drinking water
How is DI Water Produced?
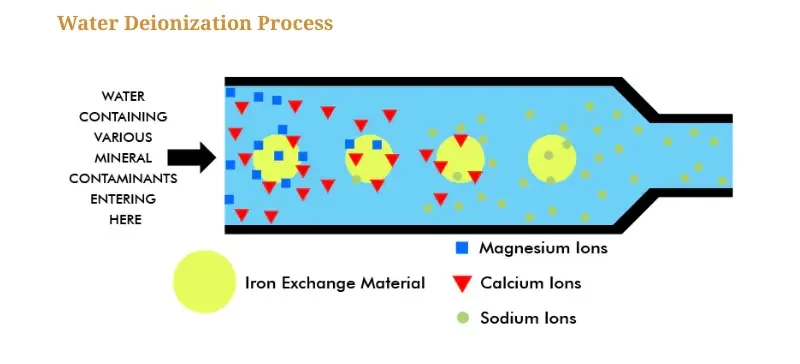
Ion Exchange:
Water is passed through resin beds that swap cations for hydrogen ions (H⁺) and anions for hydroxide ions (OH⁻), producing pure H₂O.Reverse Osmosis (RO):
A pre-treatment or standalone process that forces water through a semi-permeable membrane to remove dissolved salts, bacteria, and organic material.Distillation:
Water is boiled and the steam is condensed, leaving behind impurities. Though effective, this method is energy-intensive and slower than modern alternatives.
Applications of DI Water in the Laboratory
Deionized water is a fundamental tool in virtually all scientific disciplines, including chemistry, biology, and materials science. Its primary uses include:
Solution Preparation:
Used for making buffers, reagents, and dilutions without introducing unwanted ions.Chromatography (HPLC, GC):
Ensures uncontaminated solvents for analytical separation.Electrochemistry:
Prevents interference from stray ions in conductivity and voltage measurements.Cell Culture & Microbiology:
Provides a contaminant-free medium for preparing culture media and diluents.Cleaning and Sterilization:
Leaves no residue on glassware or instruments, avoiding cross-contamination.Spectrometry (AAS, AES):
Necessary for trace element detection at very low concentrations.Autoclaves & Analytical Devices:
Reduces scale and corrosion, ensuring long equipment life.
Benefits of Installing a DI Water System in Your Lab
Accurate and Reproducible Results:
Eliminates variability caused by contaminants, ensuring high data integrity.Operational Efficiency:
On-demand access to high-purity water improves workflow and eliminates reliance on bottled supplies.Extended Equipment Lifespan:
Reduces scaling, corrosion, and wear on precision instruments.Cost Savings Over Time:
Investing in a DI system is more economical than purchasing pre-packaged pure water.Regulatory Compliance:
Helps meet water quality standards in pharmaceutical, clinical, and food laboratories.Customization & Scalability:
Modern DI systems can be tailored for flow rate, purity level, and capacity needs.
Comparison: DI Water vs. Other Water Types
| Water Type | Purity Level | Typical Applications |
|---|---|---|
| Tap Water | Low | Basic cleaning, non-critical processes |
| Distilled Water | Medium | General reagent prep, low-sensitivity work |
| Deionized Water | High | Analytical chemistry, instrumentation, cell culture |
Common DI System Issues & Troubleshooting
| Issue | Likely Cause | Recommended Fix |
|---|---|---|
| Poor Water Quality | Expired resin, dirty filters, poor source | Replace resin/filters, pre-filter the feed |
| Low Flow Rate | Clogged filters, low pressure, pump issues | Clean or replace filters, inspect system |
| Leaks | Loose fittings, degraded tubing or valves | Tighten or replace worn components |
| Excessive Usage | Oversized system, faulty pressure valve | Adjust settings, check for hidden leaks |
Expert Tips for Choosing the Right DI System
Assess Your Needs: Consider volume, required purity, and frequency of use
Check Certifications: Look for ASTM, ISO, or similar compliance
Plan for Maintenance: Opt for systems with user-friendly service schedules
Future-Proofing: Choose a system that can scale with your lab’s growth
Conclusion
Deionized water is not just a lab utility—it’s a foundation for high-quality research and reliable scientific outcomes. By investing in a well-designed, properly maintained DI water system, laboratories can protect their experiments, extend the life of their equipment, and operate more efficiently and economically. Whether you’re in academic research, pharmaceuticals, environmental science, or industry, ensuring a consistent supply of ultra-pure DI water is one of the smartest decisions your lab can make.










