Introduction: Why Compare XRF and XRD?
X-ray-based technologies are essential tools in materials science, chemistry, geology, and industrial quality control. Two standout methods—X-ray Fluorescence (XRF) and X-ray Diffraction (XRD)—are frequently used in laboratories, but serve different analytical purposes. Understanding the difference between XRF and XRD can significantly improve the accuracy of your results and help you choose the most efficient approach.
This expert-backed guide offers a comprehensive comparison of XRF and XRD, covering their principles, advantages, applications, and ideal use cases—empowering researchers and professionals to make informed decisions.
What Is XRF (X-ray Fluorescence)?
X-ray Fluorescence (XRF) is a non-destructive analytical technique used to determine the elemental composition of materials. When a material is exposed to high-energy X-rays, its atoms become excited and emit secondary (fluorescent) X-rays, which are unique to each element.
Key Features of XRF:
Identifies and quantifies elements from parts per million (ppm) to 100%
Works with solids, powders, and even liquids
Rapid analysis—typically completed within seconds to minutes
Minimal sample preparation
Portable handheld and benchtop models available
XRF Techniques:
Energy Dispersive XRF (EDXRF): Uses solid-state detectors to analyze a broad spectrum of elements.
Wavelength Dispersive XRF (WDXRF): Employs crystals and slits to separate wavelengths, offering higher resolution for complex samples.
What Is XRD (X-ray Diffraction)?
X-ray Diffraction (XRD) is used to determine the crystal structure and phase composition of materials. When X-rays interact with the orderly atomic planes of a crystalline substance, they diffract in predictable patterns. These diffraction patterns—analyzed using Bragg’s Law—reveal critical structural details.
Key Features of XRD:
Identifies phases, polymorphs, lattice parameters, and crystallinity
Highly sensitive to structural changes and crystal defects
Ideal for powdered, thin-film, and crystalline bulk samples
Provides detailed insights into phase transformations and material stability
Technical Comparison: XRF vs XRD
Real-World Applications
| Feature | XRF | XRD |
|---|---|---|
| Primary Purpose | Elemental analysis | Structural/crystallographic analysis |
| Output Data | Elemental spectra | Diffraction pattern (2θ vs intensity) |
| Speed | Seconds to minutes | Minutes to hours |
| Sample Type | Solid, powder, liquid | Crystalline solids |
| Preparation | Minimal | Finely ground, homogeneous |
| Portability | Handheld and benchtop | Mostly benchtop instruments |
| Detection Focus | Elements (Na–U or Be–U) | Crystallographic phases, defects |
| Non-destructive | Yes | Yes |
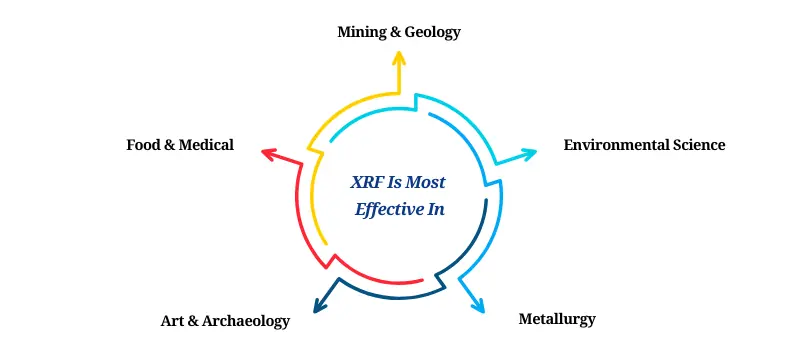
Where XRF Excels:
Mining & Geology: Rapid ore composition analysis
Environmental Science: Detecting contaminants in soil, air, and water
Metallurgy: Quality control in alloy production
Art & Archaeology: Non-invasive examination of artifacts
Food & Medical: Trace elemental detection in biological or nutritional samples
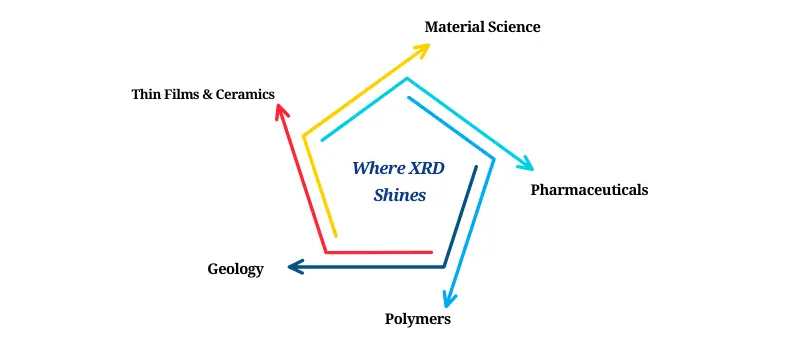
Where XRD Shines:
Material Science: Identifying phases in metals, ceramics, and composites
Pharmaceuticals: Detecting polymorphs and assessing stability
Polymers: Understanding crystallinity and phase transitions
Geology: Mineral identification in rocks and sediments
Thin Films & Ceramics: Phase evolution and texture analysis
Strengths and Limitations
✅ XRF Advantages:
Quick, quantitative results
Effective across a wide range of sample types
Portable instruments available for fieldwork
❌ XRF Limitations:
Less accurate for light elements (e.g., Be, B)
Affected by matrix effects
Requires standards for precise quantification

✅ XRD Advantages:
High sensitivity to crystalline structure and phase purity
Ideal for identifying polymorphs and complex materials
Suitable for stress/strain and texture analysis
❌ XRD Limitations:
Not suitable for amorphous materials
Requires well-prepared, homogeneous samples
Limited in trace element detection
Complementary Use of XRF and XRD
While XRF and XRD differ in focus—composition vs structure—they are often used together for complete material characterization. For instance, XRF can identify that a sample contains calcium and iron, while XRD can determine whether these elements form CaCO₃, CaO, or Fe₂O₃.
Modern lab systems increasingly integrate both techniques into a single platform, allowing users to extract comprehensive data from a single sample.
Clarifying Common Misconceptions
XRF is not suitable for crystal structure analysis.
XRD cannot determine elemental concentration directly.
WDXRF crystals are not to be confused with crystalline samples analyzed by XRD.
Additionally, EDS (Energy Dispersive Spectroscopy), often used with SEM, is similar to XRF but uses electron beams and has different strengths—especially useful for localized elemental analysis.

Conclusion: XRF or XRD—Which Should You Choose?
Use XRF when you need fast, non-destructive elemental analysis.
Use XRD when you need precise structural information about crystalline materials.
Use both for complete insight into your sample’s composition and structure.
The future of materials characterization lies in multi-technique integration, and understanding the strengths of each method will help you make the best decision.
📞 Need Help Selecting the Right Instrument?
Contact AELAB today for expert advice and customized solutions in XRF and XRD analysis.

























