Introduction
A pH meter is an essential instrument used across various industries to accurately measure the acidity or alkalinity of a solution. Whether you’re in a research lab, wastewater treatment plant, brewery, or on a farm, understanding pH values is critical to quality control, safety, and scientific analysis.
In this complete guide, we’ll cover how pH meters work, where they’re used, the types available, and how to choose the best one for your needs.

What Is a pH Meter?
A pH meter is a scientific device that measures hydrogen ion concentration ([H⁺]) in a water-based solution. The measurement is expressed as pH, which ranges from 0 (very acidic) to 14 (very alkaline), with 7 considered neutral.
pH stands for “potential of hydrogen” and is a logarithmic scale used to indicate how acidic or basic a solution is.
How Does a pH Meter Work?
A typical device includes three key components:
Internal Electrode (Ag/AgCl wire)
Reference Electrode (usually similar materials)
High-impedance Voltmeter
These are often combined in a glass electrode probe. Inside the probe is a buffer solution (usually pH 7). When the probe is immersed in a sample, it measures the voltage difference between the internal buffer and the sample. This voltage is then converted into a pH value and displayed on the meter.
Accurate pH measurement requires regular calibration using standardized buffer solutions (e.g., pH 4.0, 7.0, 10.0).

What Does a pH Meter Measure?
A pH meter measures the activity of hydrogen ions in a solution, helping you determine whether the solution is:
Acidic (pH < 7)
Neutral (pH = 7)
Alkaline/Basic (pH > 7)
It provides more precise and reliable results than traditional test strips or indicators and is widely used where accuracy is vital.
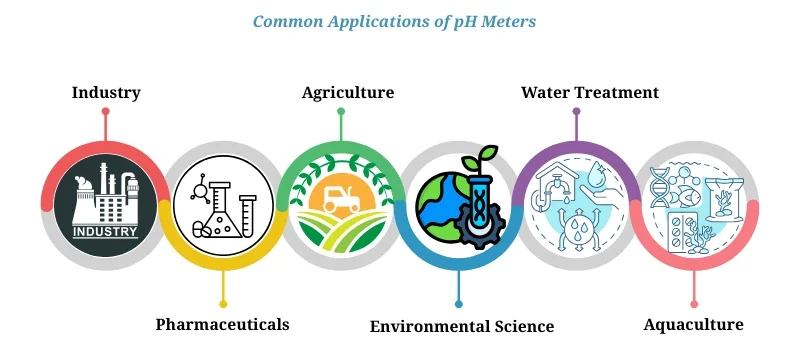
Common Applications
pH meters are incredibly versatile and are used in various fields including:
| Industry | Application |
|---|---|
| Food & Beverage | Quality control, fermentation monitoring, beverage acidity testing |
| Pharmaceuticals | Ensuring drug safety, pH monitoring in formulation and production |
| Agriculture | Soil pH testing for nutrient availability and crop optimization |
| Environmental Science | Water quality testing in rivers, lakes, and oceans |
| Water Treatment | Monitoring acidity in municipal and industrial water systems |
| Aquaculture | Maintaining stable pH levels in aquariums and fish farms |
Types of pH Meters
Depending on your application, you can choose from several types of pH meters:
Pocket pH Meters
Portable, compact, and budget-friendly
Ideal for fieldwork, aquariums, or quick testing
Limitations: Less accurate, fewer features
Benchtop pH Meters
Larger, more accurate, and lab-grade
Often feature advanced software and multi-point calibration
Best for: Laboratories, research, and industrial quality control
Specialty Meters
Designed for extreme conditions (e.g., high temperatures, low-ion samples)
Include flat-surface, non-clog, or antimony electrodes
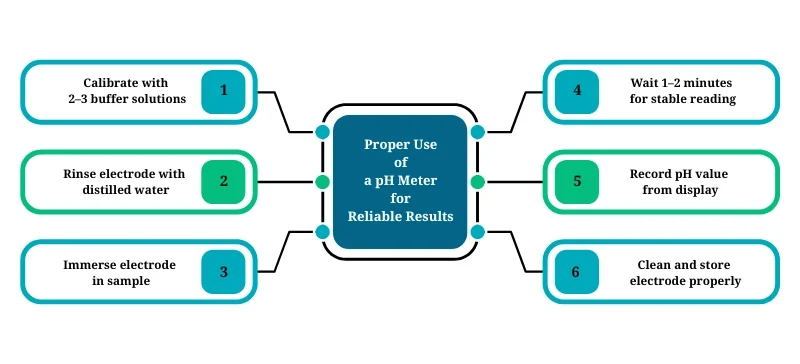
How to Use a pH Meter Properly
Follow these steps to ensure accurate and consistent readings:
Calibrate your pH meter with buffer solutions (at least 2 points, ideally 3)
Rinse the electrode with distilled water before use
Immerse the electrode in your sample
Wait 1–2 minutes for the reading to stabilize
Record the value shown on the display
Clean the electrode after each use and store it in storage solution
Choosing the Right pH Meter: 4 Key Factors
1. Accuracy
Choose a pH meter with the appropriate level of accuracy for your needs:
±0.01 pH: Suitable for general lab and industrial use
±0.001 pH or better: Needed for research-grade applications
Tip: Even minor inaccuracies can drastically impact results in fields like pharmaceuticals or biotechnology.
2. Resolution
Resolution reflects the smallest change your meter can detect.
Common: 0.01 pH
High-sensitivity applications: 0.001 or 0.0001 pH
3. Electrode Compatibility
Select an electrode based on your sample type:
Glass Bulb: Great for most aqueous solutions
Polymer/Non-Clog Tips: Ideal for food, viscous, or semi-solid samples
Flat-Cell Electrodes: Portable and durable for fieldwork
4. Long-Term Usability
Consider:
Ease of calibration
Replacement electrode cost
Software/data connectivity
Durability and waterproofing for field units
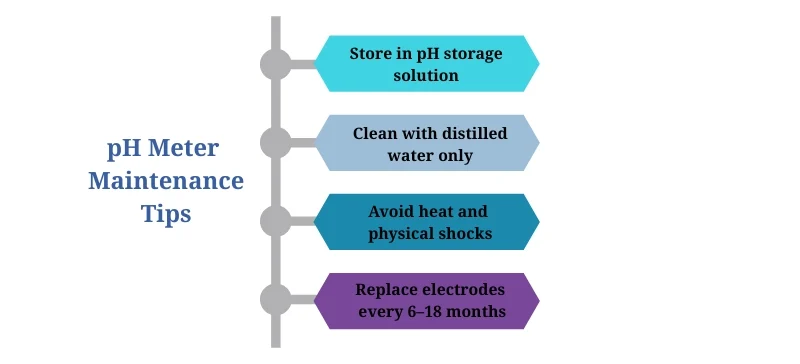
Maintenance Tips
Proper care extends your meter’s lifespan and accuracy:
Store electrodes in pH storage solution, never dry
Clean with distilled water, never tap water
Avoid extreme temperatures or mechanical shocks
Replace electrodes periodically (typically every 6–18 months)
Final Thoughts
Whether you’re monitoring water quality, testing soil, formulating medicine, or brewing craft beer, a high-quality pH meter is essential. By understanding how these devices work and selecting the right model for your needs, you can ensure accuracy, reliability, and long-term performance in your measurements.











