Introduction
Autoclaves (steam sterilizers) are essential in laboratories and clinical settings for decontaminating instruments, glassware, and biohazardous waste. Because they use high-pressure saturated steam, autoclaves present thermal, pressure, and biological risks if not used correctly. This autoclave safety guide explains the science behind steam sterilization, safe operating practices, monitoring and maintenance, and emergency steps — written for lab managers, technicians, and researchers who need clear, publication-ready guidance.
How steam sterilization works (the science)
Steam sterilization is the core process described in every autoclave safety guide. Autoclaves use saturated steam under pressure to efficiently transfer thermal energy into the load. The steam condenses on cooler surfaces, releasing latent heat that quickly kills microorganisms.
Typical validated sterilization parameters are 121°C at ~15 psi, maintained for a validated exposure time once the chamber and load reach target temperature. Higher temperatures, such as 134°C, are sometimes used for shorter instrument cycles.
Effective sterilization requires full steam contact with all surfaces — air pockets, overpacked loads, or impermeable wraps can block steam flow and reduce effectiveness.
Major hazards and required personal protective equipment (PPE)
As every autoclave safety guide emphasizes, autoclaves can pose several risks if used carelessly:
Thermal risks: burns from steam, hot surfaces, and autoclaved liquids.
Mechanical/pressure risks: injury from improperly sealed containers or damaged door gaskets; risk of exploding containers.
Biological risks: exposure from improperly contained infectious materials.
Chemical/radioactive risks: some chemicals and radionuclides should never be autoclaved.
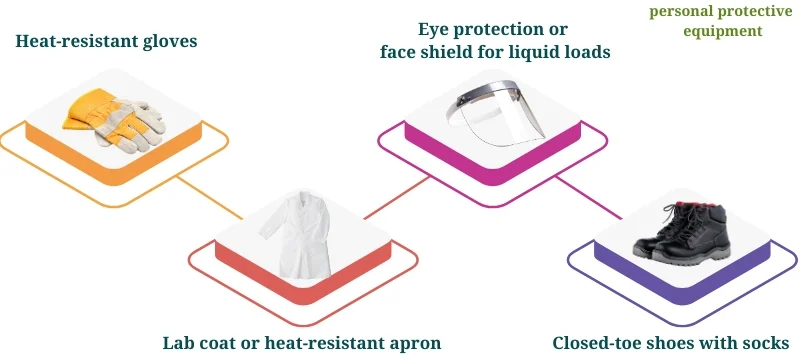
Minimum PPE when loading/unloading:
Heat-resistant gloves (and cut-resistant gloves when handling sharps)
Lab coat or heat-resistant apron
Eye protection or face shield for liquid loads
Closed-toe shoes with socks
Preparing materials for autoclaving
General packing principles
Pack loosely to allow steam circulation; do not overfill bags or trays.
Use autoclavable trays (stainless steel or autoclave-safe plastics) to contain leaks and make handling safer.
Check glassware for cracks; use borosilicate glass for liquid loads.
Open or loosen caps on liquid containers to prevent pressure buildup.
What not to autoclave
Flammable, volatile, or corrosive chemicals (alcohols, solvents, concentrated acids).
Radioactive materials (follow radiation safety rules).
Certain pathological samples, low-molecular-weight toxins, and prions may require specialized treatment — consult biosafety officers.
Choosing the right cycle: gravity vs. liquid vs. preprogrammed
Autoclave Cycle Types and Typical Parameters
| Cycle Type | Typical Use | Key Characteristics | Typical Temperature & Exposure Time |
|---|---|---|---|
| Gravity (Air-Displacement) | Wrapped instruments, glassware, porous materials | Steam displaces air from the chamber; efficient for solid or porous loads | 121 °C for ≥15 min |
| Liquid Cycle | Media, solutions, and liquid samples | Slower depressurization to prevent boil-over; extended drying time | 121 °C for 30–60 min (depending on volume) |
| Preprogrammed “Waste” Cycle | Biohazardous waste and disposable lab materials | Automated sterilization and venting; must be validated for load type | 121–134 °C for 30–60 min total process time |
Tip:
Cycle time should always be adjusted based on load density, packaging, and chamber validation. Heavily packed or infectious loads typically require longer exposure and verified biological indicator testing.
Step-by-step safe operating procedure
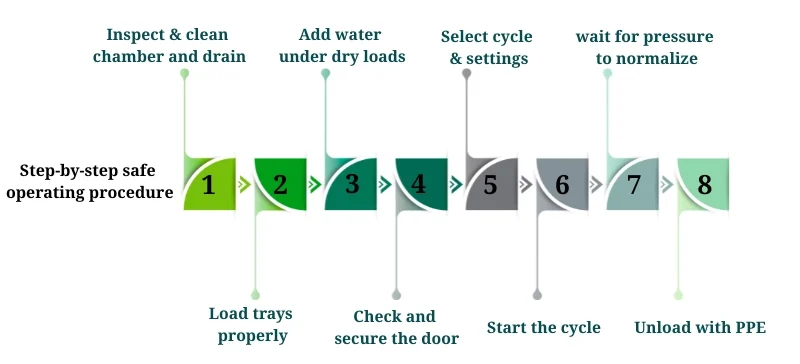
Inspect chamber and drain strainer; remove foreign objects from previous users.
Pack trays so steam can circulate; avoid direct contact with chamber floor.
Add small amounts of water under dry loads or place liquid containers in trays with ~1 inch of water to reduce thermal shock.
Close and latch door; confirm gasket condition.
Select appropriate cycle (gravity/liquid/dry) and validate settings:
Target temp (commonly 121°C)
Exposure time (based on load)
Drying option as needed
Start cycle and log the run if your facility requires it.
After cycle completion, wait for pressure to equalize and allow the chamber to cool partially before opening.
Open door slowly, stand to one side, and allow steam to vent.
Use PPE to remove loads; allow liquids to cool further (recommended: 10–60 minutes depending on load).
Monitoring, validation, and recordkeeping
Routine load indicators
Use heat-sensitive indicator tape or internal pack indicators for every load. These show that a target temperature was reached but do not prove sterilization (they don’t confirm time/pressure exposure).
Biological indicators & validation
Periodically (commonly monthly or per institutional SOP) run biological indicators containing heat-resistant spores (e.g., Geobacillus stearothermophilus) placed at the most challenging location in a load to confirm the cycle lethality.
Maintain records of biological indicator results, cycle printouts (time, temperature, pressure), and any corrective actions.
Documentation
Keep logs for preventive maintenance, repairs, training, and cycle records accessible to users and safety personnel.
Maintenance, troubleshooting, and what to do on failure
The autoclave safety guide emphasizes regular maintenance to ensure reliable sterilization. Always follow the manufacturer’s schedule and let only qualified technicians service the unit.
If a unit malfunctions:
Stop use immediately and post an “Out of Service” notice.
Notify your facilities/safety contact and the service provider.
Do not attempt major repairs unless trained and authorized.
Common problems: clogged drains, worn door gaskets, incomplete steam supply, and recorder failures — all affect sterilization reliability.
Emergency procedures
Quick and calm response to autoclave incidents—such as burns, spills, or equipment failures—can prevent serious injuries and maintain lab safety
Burns: Immediately cool the burn with cool (not ice) running water for at least five minutes; seek medical attention for facial, large, or blistering burns. Report occupational injuries per institution protocol.
Spills inside autoclave: Allow the unit and contents to cool to room temperature before cleanup. Use appropriate spill kits and remove sharps with tongs or a brush and pan.
Explosions or significant mechanical damage: Evacuate the area and contact emergency services and your institutional safety office.

Roles & responsibilities (summary)
Clear role definition ensures accountability and consistent autoclave safety—each team member plays a key part in maintaining compliance and preventing accidents.
Lab supervisors/Principal Investigators: Ensure personnel receive documented hands-on training and that autoclave use follows institutional policies.
Department/facility managers: Maintain service contracts and ensure qualified maintenance.
Autoclave operators: Follow SOPs, use PPE, maintain logs, and report malfunctions or incidents.
Benefits of following autoclave safety best practices
Following autoclave safety best practices enhances lab safety, ensures sterilization reliability, and reduces long-term operational risks.
Protects personnel from burns and biological exposure.
Ensures reliable sterilization and experimental integrity.
Reduces regulatory and waste-disposal complications.
Extends equipment life and lowers repair costs.
Conclusion
Maintaining proper autoclave safety practices is essential for every laboratory. By following this autoclave safety guide, staff can safely operate sterilizers, prevent accidents, and guarantee complete decontamination of materials.
A well-maintained autoclave not only protects personnel but also preserves research quality and compliance with safety regulations. Safe autoclave use is a cornerstone of responsible, high-quality laboratory work.
For best results, always train new users, document maintenance activities, and verify sterilization cycles using biological indicators. Safe autoclave use is not just a protocol—it’s a commitment to responsible science and a safer working environment.
Take Action for Safer Lab Practices
Ensure your laboratory operates safely and efficiently by implementing these autoclave safety practices today. Train your staff, maintain your equipment, and validate every cycle to protect personnel, ensure proper sterilization, and comply with safety regulations.
Call to Action :
Explore AELAB’s full range of autoclaves, trays, and monitoring solutions to upgrade your lab’s sterilization capabilities. Stay informed, stay compliant, and make safety a top priority in every experiment.












