ntroduction
In the pharmaceutical and medical device industries, maintaining high standards of quality control, safety, and regulatory compliance is essential. Analytical techniques such as Gas Chromatography (GC) and High-Performance Liquid Chromatography (HPLC) play a critical role in achieving these goals. This article presents a comprehensive GC vs. HPLC comparison, focusing on how these powerful chromatographic methods enable precise separation, identification, and quantification of complex mixtures to ensure the safety, efficacy, and reliability of pharmaceutical products and medical devices.
The accuracy of analytical testing directly impacts patient safety and product performance.Therefore, both GC and HPLC are widely used to verify product composition, detect impurities, and ensure compliance with international regulatory standards. Moreover, understanding the strengths, limitations, and appropriate applications of each technique is essential for selecting the most effective analytical approach in pharmaceutical and medical device testing.

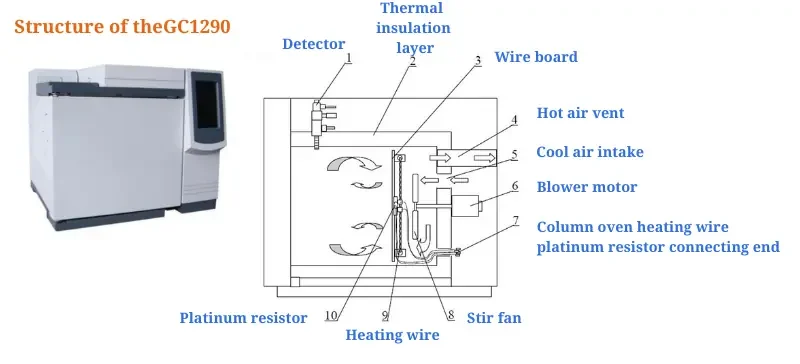
Gas Chromatography (GC) in Pharmaceutical and Medical Device Testing What is GC?
In general, Gas Chromatography (GC) is a widely used technique for analyzing volatile and semi-volatile compounds. Specifically, it separates components between a stationary phase and a mobile gas phase, such as helium or nitrogen. Therefore, GC is ideal for substances that can be vaporized without thermal decomposition.
Key Applications of GC in Pharmaceutical Testing:
Residual Solvent Analysis: GC plays a vital role in detecting and quantifying residual solvents in pharmaceutical products, ensuring compliance with safety standards.
Impurity Detection: GC is used to identify and quantify trace amounts of impurities that could affect drug quality and patient safety.
Drug Formulation Analysis: GC is essential in analyzing pharmaceutical formulations, ensuring that active pharmaceutical ingredients (APIs) are present in the correct concentrations.
Stability and Pharmacokinetic Studies: Additionally, GC helps monitor drug stability and track absorption, distribution, metabolism, and excretion (ADME) in biological systems.

Applications in Medical Device Testing:
Material Characterization: GC analyzes the chemical composition of materials used in medical devices to ensure safety and biocompatibility.
Sterilization Validation: For devices undergoing sterilization processes, GC ensures that no harmful residues remain post-sterilization.
Residual Solvent Analysis: Similar to its role in pharmaceuticals, GC helps detect and quantify residual solvents in medical device materials.

High-Performance Liquid Chromatography (HPLC) in Pharmaceutical Testing What is HPLC?
Key Applications of HPLC in Pharmaceutical Testing:
Drug Quantification and Purity Testing: HPLC is used extensively for quantitative analysis of APIs and excipients, ensuring they meet regulatory standards for purity and concentration.
Impurity and Degradation Product Analysis: HPLC is crucial for identifying and quantifying degradation products and other impurities, ensuring the safety and stability of pharmaceutical products.
Therapeutic Drug Monitoring:
HPLC is used to measure drug concentrations in biological fluids such as plasma and serum. As a result, it helps ensure proper therapeutic levels and improves patient safety.
Stability Testing: HPLC is used to assess how drugs degrade over time and under various environmental conditions, ensuring that products maintain their potency and efficacy.
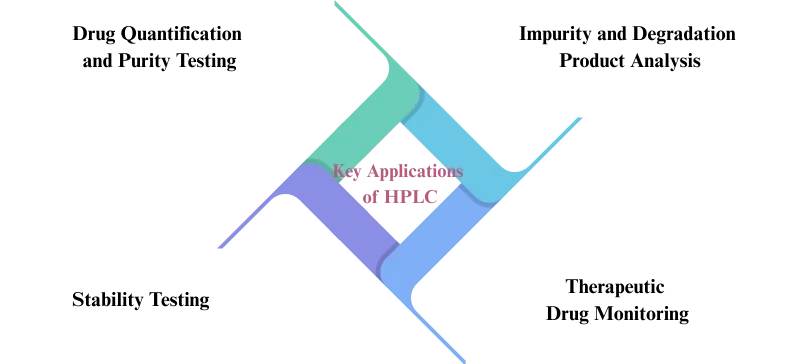
Comparison of GC vs. HPLC: Choosing the Right Technique
From a GC vs. HPLC perspective, each technique provides unique advantages based on the analyte characteristics and the type of analysis being performed.
| Feature | High-Performance Liquid Chromatography (HPLC) | Gas Chromatography (GC) |
|---|---|---|
| Mobile Phase | Liquid (mixture of solvents and water) | Gas (helium, nitrogen, hydrogen) |
| Sample State | Non-volatile, thermally unstable, large molecules | Volatile or derivatizable compounds |
| Separation Principle | Partitioning, adsorption, ion exchange, size exclusion | Based on boiling point and polarity |
| Detector Types | UV-Vis, Refractive Index, Fluorescence, MS | Flame Ionization, TCD, ECD, MS |
| Typical Applications | APIs, excipients, biomolecules, chiral separations | Residual solvents, volatile impurities |
| Coupling with MS | HPLC-MS for peptides, metabolites, biologics | GC-MS for residual solvents, volatile impurities |
Synergy Between GC and HPLC: A Comprehensive Approach
While both GC and HPLC serve distinct roles, laboratories often use them together in modern pharmaceutical and medical device testing. Moreover, when laboratories combine these techniques with Mass Spectrometry (MS), they perform a more comprehensive analysis of complex samples and achieve higher accuracy and improved reproducibility.
For example, GC effectively analyzes volatile compounds; however, HPLC better handles non-volatile and thermally unstable substances. Therefore, by integrating both techniques, laboratories generate more comprehensive analytical results and improve sensitivity, specificity, and overall data reliability.
Challenges and Future Trends in GC and HPLC
Despite their widespread use, both GC and HPLC face challenges in analytical testing, such as:
Matrix Interference: Complex samples introduce interfering components and directly reduce result accuracy.
Sensitivity and Detection Limits: Some analytical methods struggle to detect trace-level compounds.
Sample Complexity: Complex samples require advanced techniques or longer analysis times.
Future Trends
- Column Technology Improvements: New column designs shorten analysis time and enhance separation efficiency.
- Detector Sensitivity: Advanced detectors increase sensitivity and improve resolution in both GC and HPLC methods.
- Automation and AI Integration: AI and automation streamline data analysis, boost efficiency, and minimize human error in analytical testing.

Conclusion: The Essential Role of GC and HPLC in Pharmaceutical and Medical Device Testing
Overall, GC vs. HPLC is a key consideration in pharmaceutical and medical device testing, as both Gas Chromatography (GC) and High-Performance Liquid Chromatography (HPLC) play vital roles in ensuring product safety, efficacy, and regulatory compliance. Moreover, by understanding their respective strengths, limitations, and technological advancements, laboratories can therefore optimize analytical workflows and achieve more accurate, reliable, and efficient testing outcomes.
Call to Action
Therefore, if you are evaluating GC vs. HPLC for pharmaceutical or medical device testing, AELAB offers expert consultation and optimized analytical solutions tailored to your needs. Moreover, our experienced team helps you achieve highly accurate results while simultaneously ensuring full compliance with international regulatory standards.