Introduction
Ion Chromatography is a powerful analytical technique used to separate and quantify charged species in complex samples. Due to growing regulatory demands in environmental, pharmaceutical, food, and industrial analysis, IC has become essential for accurate and reliable ion detection at trace levels.
This article provides a clear overview of the fundamental principles, operating mechanisms, classifications, and major applications of this analytical technique, enabling professionals and laboratory specialists to better understand its scientific significance and practical value.
What Is Ion Chromatography?
Ion Chromatography (IC), also known as Ion-Exchange Chromatography (IEC), is a liquid chromatography technique designed to separate, identify, and quantify positively and negatively charged ions in a sample.
Principle of Ion Chromatography
The fundamental basis of this analytical technique is ion exchange, a reversible electrostatic interaction between charged analytes and oppositely charged functional groups bound to the stationary phase.
Ion Exchange Mechanism Explained
Inside the IC column:
The stationary phase consists of resin beads.
These beads are functionalized with charged groups.
Oppositely charged ions in the sample are retained.
Each ion interacts with different strength.
Separation occurs based on retention time differences.
Example:
In anion-exchange chromatography, negatively charged ions (Cl⁻, NO₃⁻, SO₄²⁻) are retained by positively charged resin.
In cation-exchange chromatography, positively charged ions (Na⁺, K⁺, Ca²⁺) bind to negatively charged resin.
The stronger the ionic interaction, the longer the retention time.
How Does Ion Chromatography Work? (Step-by-Step Process)
Understanding the IC workflow improves method development and troubleshooting.
1. Sample Preparation
Filtration (0.22 µm or 0.45 µm)
Dilution if required
Removal of particulates
Extraction for solid samples
Proper preparation prevents column contamination and ensures reproducibility.
2. Sample Injection
The prepared liquid sample is introduced into the mobile phase stream.
3. Eluent Flow
A high-pressure pump carries the sample through the column using an eluent.
4. Ion Separation
Ions interact with the stationary phase and separate based on:
Charge
Size
Affinity
Ionic strength
pH conditions
5. Suppression (in Suppressed IC Systems)
A suppressor reduces background conductivity, dramatically increasing sensitivity and lowering detection limits.
6. Detection
Most systems use conductivity detection, although advanced systems may integrate:
UV detection
Electrochemical detection
IC-MS (Ion Chromatography–Mass Spectrometry)
7. Data Analysis
Each ion appears as a chromatographic peak:
Retention time → identifies the ion
Peak area → determines concentration
Modern IC software enables automated calibration, quantification, and compliance reporting.
What Does Ion Chromatography Measure?
IC measures both inorganic and organic ions.
Common Anions:
Fluoride (F⁻)
Chloride (Cl⁻)
Nitrate (NO₃⁻)
Nitrite (NO₂⁻)
Sulfate (SO₄²⁻)
Phosphate (PO₄³⁻)
Bromide (Br⁻)
Common Cations:
Sodium (Na⁺)
Potassium (K⁺)
Calcium (Ca²⁺)
Magnesium (Mg²⁺)
Ammonium (NH₄⁺)
Lithium (Li⁺)
Manganese (Mn²⁺)
These ions are critical in environmental regulation, pharmaceutical quality, industrial production, and clinical testing.

Types of Ion Chromatography
1. Anion-Exchange Chromatography
-
Stationary phase: Positively charged
-
Retains: Negatively charged ions
-
Widely used in water and environmental testing
2. Cation-Exchange Chromatography
-
Stationary phase: Negatively charged
-
Retains: Positively charged ions
-
Common in pharmaceutical and industrial analysis
3. Suppressed vs Non-Suppressed IC
| Feature | Suppressed IC | Non-Suppressed IC |
|---|---|---|
| Sensitivity | Very High | Moderate |
| Background Conductivity | Low | Higher |
| Complexity | Moderate | Simple |
| Best For | Trace-level analysis | High concentration samples |
4. Ion-Exclusion Chromatography
Used primarily for weak acids and neutral polar molecules.
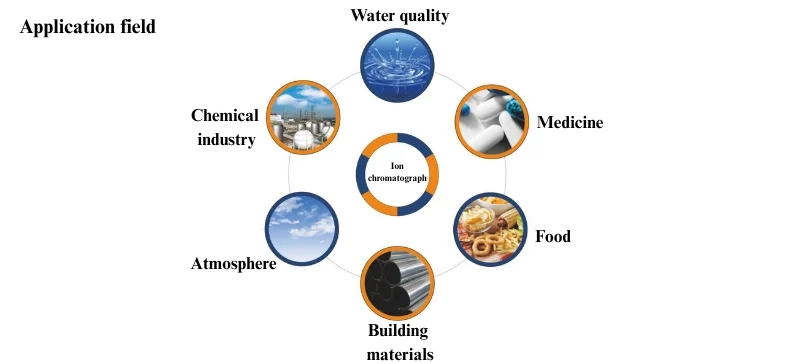
Applications of Ion Chromatography
Ion Chromatography is indispensable across industries.
1. Environmental and Water Analysis
Drinking water quality control
Wastewater monitoring
Groundwater nitrate analysis
Fluoride and sulfate measurement
Compliance with EPA, ISO, WHO standards
2. Pharmaceutical Industry
Counterion analysis
Impurity profiling
Stability testing
Dissolution studies
USP-compliant ion testing
Accurate ion quantification directly impacts drug safety and regulatory approval.
3. Food and Beverage Industry
Salt content determination
Organic acid analysis
Sugar alcohol testing
Preservative monitoring
Mineral water quality control
4. Industrial Applications
Electroplating bath analysis
Semiconductor manufacturing
Catalyst monitoring
Corrosion and scaling analysis
Brine and raw material testing
5. Clinical and Biological Research
Electrolyte testing in serum and urine
Porphyrin analysis
HbA1c and enzyme studies
Kidney dialysis monitoring
Ion Chromatography vs HPLC
| Feature | Ion Chromatography | HPLC |
|---|---|---|
| Separation Mechanism | Ionic interactions | Hydrophobic/polar interactions |
| Target Compounds | Inorganic & ionic species | Organic molecules |
| Detector | Conductivity | UV/Vis |
| Best Use | Water & ion analysis | Drug & organic analysis |
Professional Tips for Optimizing IC Performance
From practical laboratory experience:
Always filter samples before injection.
Use freshly prepared eluents.
Maintain stable column temperature.
Regularly calibrate with certified standards.
Monitor suppressor health.
Document system suitability parameters.
Avoid overloading the column.
These practices ensure analytical accuracy and regulatory compliance.
Historical Development
1940s: Ion exchange used for rare earth separation
1975: Term “Ion Chromatography” introduced
1980s: Suppressed conductivity systems developed
Today: IC-MS coupling and multi-channel systems enable advanced speciation analysis
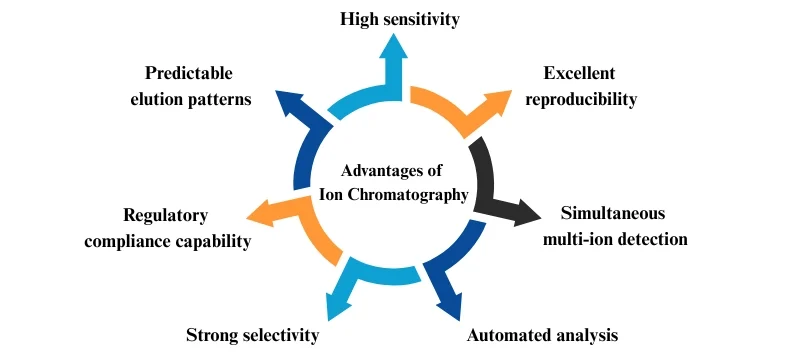
Advantages of Ion Chromatography
- High sensitivity (ppb detection limits)
- Excellent reproducibility
- Simultaneous multi-ion detection
- Automated analysis
- Strong selectivity
- Regulatory compliance capability
- Predictable elution patterns
Compared to traditional wet chemistry methods, IC provides faster, more reliable, and cost-efficient analysis.
Limitations of Ion Chromatography
- Limited to ionizable compounds
- Requires pH control
- Column lifetime considerations
- Matrix interference in complex samples
- Moderate run time (typically 30–60 minutes)
Proper training and preventive maintenance significantly improve system longevity and performance.
The Future of Ion Chromatography
Modern innovations include:
Capillary IC systems
Automated multi-ion platforms
IC coupled with ICP-MS
Smart software integration
Miniaturized systems for on-site analysis
As environmental regulations and pharmaceutical standards continue to tighten, the demand for precise ion analysis will grow.
Ion Chromatography remains a cornerstone of analytical chemistry laboratories worldwide.
Conclusion
Ion Chromatography (IC) is a highly accurate, sensitive, and versatile analytical technique for ionic analysis across environmental, pharmaceutical, food, industrial, and clinical applications.
Its foundation in ion-exchange chemistry enables predictable, selective, and reproducible separations. With advancements such as IC-MS integration, capillary systems, and intelligent automation, IC continues to expand its capabilities in modern laboratories.
For laboratories seeking precision, compliance, and long-term analytical reliability, Ion Chromatography remains an essential investment.