Spectrophotometer vs. Fluorometer: Key Differences and When to Use Each:Spectrophotometers and Fluorometers are crucial analytical tools for analyzing light interactions with samples in scientific research. Spectrophotometers measure absorbed light and are ideal for general analyses, while fluorometers detect emitted fluorescence with high sensitivity, making them suitable for detecting compounds at low concentrations. These instruments are essential in both routine and specialized applications, including environmental and biochemical studies.
Key Differences Between Spectrophotometers and Fluorometers
Measurement Principle
- Spectrophotometer: Measures light absorption by a sample. A light beam passes through the sample, and the instrument quantifies how much light is absorbed at specific wavelengths. Based on the Beer-Lambert Law, this absorption is proportional to the concentration of the substance.
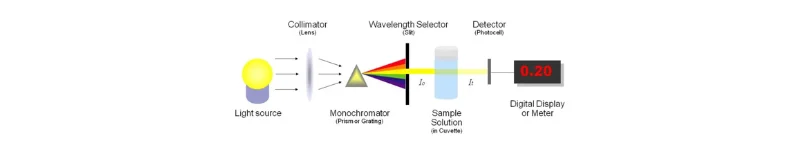
Figure 1:
Diagram of spectrophotometer components, including the light source, monochromator, and sample cuvette, illustrating the process of light absorption measurement.
- Fluorometer: Detects fluorescence emitted by a sample. After a light source excites certain molecules, they emit light at different wavelengths. The instrument measures this emission, which corresponds directly to the concentration of the fluorescent compound.
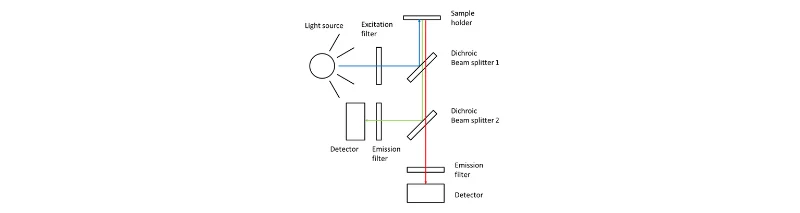
Figure 2:
Illustration of a fluorometer setup, showing excitation and emission filters and the detector used to measure fluorescence.”
- Sensitivity and Detection Range
- Spectrophotometer: Has a broad detection range across UV, visible, and sometimes near-infrared (NIR) wavelengths, accommodating a wide variety of concentrations in liquid, solid, or gas samples.
- Fluorometer: Offers superior sensitivity for detecting trace compounds at very low concentrations (picomolar to nanomolar levels). It is designed specifically for samples with fluorescent properties, making it ideal for measuring compounds present in low amounts.
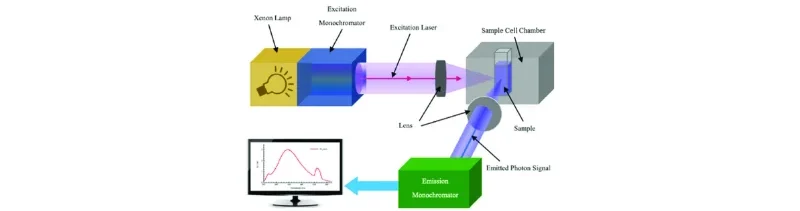
Figure 3:
Sample Requirements and Preparation
- Spectrophotometer: Requires less preparation and is suitable for diverse samples, including colored solutions and non-fluorescent compounds.
- Fluorometer: Generally requires samples to be tagged with fluorescent dyes. Conditions such as pH, temperature, and solvent must be precisely controlled to ensure consistent fluorescence, adding complexity to sample preparation.
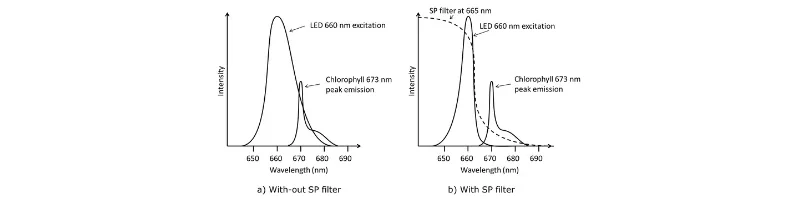
Cost and Complexity
- Spectrophotometer: Typically more affordable and easier to operate, with fewer specialized components. It is accessible for routine lab applications.
- Fluorometer: More costly due to advanced optical components required for high sensitivity. Operators need specialized training to use fluorometers accurately.
Applications and Ideal Usage Scenarios :Spectrophotometer vs. Fluorometer
- Spectrophotometer: Commonly used for general absorbance measurements, concentration analysis, and purity testing in DNA, RNA, and protein research. It is cost-effective and versatile, making it suitable for routine analyses in standard laboratories.
- Fluorometer: Ideal for detecting low-concentration analytes in molecular biology, biochemistry, and environmental sciences. It is widely used for studies involving DNA, RNA, or proteins labeled with fluorescent markers and for precise, trace-level detection.
Comparison Table: Spectrophotometer vs. Fluorometer
| Feature | Spectrophotometer | Fluorometer |
|---|---|---|
| Measurement Principle | Absorbance (Beer-Lambert Law) | Emitted fluorescence |
| Sensitivity | Moderate, suited for moderate to high concentrations | High, suited for low concentrations |
| Detection Range | Broad (UV-VIS-NIR) | Narrow, optimized for fluorescent compounds |
| Sample Requirements | Minimal preparation, diverse sample types | Requires fluorescent samples, sensitive to environmental factors |
| Cost | Generally affordable, simpler to operate | More expensive, requires specialized training |
| Applications | Routine analyses, concentration measurements, purity testing | Low-concentration detection, fluorescence assays |
Choosing the Right Instrument
Your choice between a spectrophotometer and a fluorometer depends on your laboratory needs:
- Use a Spectrophotometer if your focus is on absorbance measurements, routine concentration analysis, or when sample versatility is essential.
- Opt for a Fluorometer if high sensitivity for low-concentration detection, such as DNA or protein quantification with fluorescent markers, is crucial for your application.
Recommended AELab Products
AELab Fluorometers
For sensitive laboratory applications that require high precision in fluorescence assays, AELab offers a range of Fluorometers, including models such as,Fluorometer FD-100 ،Fluorometer Fluo Series ، Fluorometer ,. These devices are ideal for detecting fluorescent compounds in biological, biochemical, and environmental research, providing high sensitivity for trace compound detection.
AELab Spectrophotometers
AELab offers a range of spectrophotometers, including models such as the Double Beam UV-VIS Spectrophotometer AE-S80 AE-S90 Series, UV-VIS Spectrophotometer AE-S90-MD Touch, and UV-VIS Spectrophotometer AE-S80-TS/AE-S90-TD. These models are optimized for absorbance measurements in various laboratory applications, making them ideal for concentration analysis, purity testing, and routine DNA, RNA, and protein research. AELab spectrophotometers provide reliable and accurate results to support scientific advancements.

Conclusion
In conclusion, spectrophotometers and fluorometers each offer distinct advantages for analytical tasks. Spectrophotometers are versatile and economical for routine absorbance measurements, while fluorometers provide unmatched sensitivity, essential for trace analyses. By understanding their unique features, researchers can make informed choices, enhancing data accuracy and efficiency in their respective fields.

















