Introduction
A flame photometer , also known as a flame atomic emission spectrometer, is an essential analytical instrument used to determine the concentration of alkali and alkaline earth metals. It is widely applied in clinical laboratories, environmental monitoring, agriculture, food safety, and industrial quality control. The reliability and precision of flame photometric analysis depend on various operational, instrumental, and environmental factors. This comprehensive guide covers the working principle, key influencing parameters, optimization strategies, and common troubleshooting techniques—particularly focusing on AELAB flame photometers.
What Is Flame Photometry and How Does It Work?
Fundamentally, a flame photometer operates on the principle of atomic emission spectroscopy. In this process, it uses a flame as the excitation source, and subsequently, a photoelectric detection system measures the light intensity emitted by the excited atoms.
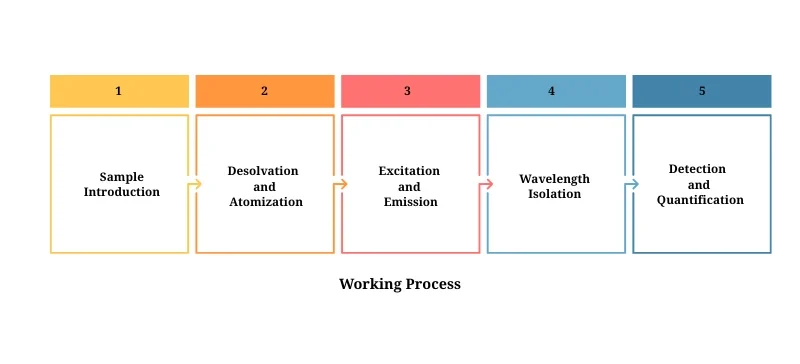
Working Process Overview:
Sample Introduction: Liquid sample is aspirated and nebulized into the flame.
Desolvation and Atomization: Solvent evaporates; metal atoms are released.
Excitation and Emission: Atoms emit radiation at specific wavelengths.
Wavelength Isolation: A monochromator selects the emission wavelength of interest.
Detection and Quantification: A photodetector measures light intensity, which is compared to a calibration curve for quantification.
Key Factors Affecting Flame Photometer Performance
1. Flame and Excitation Conditions
Flame Temperature:
Too low: Reduced atomization and sensitivity.
Too high: Ionization of alkali metals, causing non-linear responses.
Optimization: Maintain consistent fuel-to-oxidant ratio for a stable ~1900°C flame.
Gas Pressure and Flow Rate:
Unstable pressure leads to signal fluctuation.
Best Practice: Keep gas pressure around 0.12–0.15 MPa using stable gas regulators.
Sample Introduction Position:
Inconsistent burner height affects emission intensity.
Solution: Standardize the burner height and sample aspiration level.
2. Sample Properties and Composition
Ionization and Self-Absorption:
Elements like potassium may exhibit self-absorption at high concentrations.
Tip: Use ionization suppressors (e.g., cesium or lanthanum salts) and dilute samples appropriately.
Matrix Effects and Coexisting Ions:
Other alkali metals can enhance or interfere with emission lines.
Solution: Use matrix-matched standards or internal standard calibration.
Physical Characteristics:
Viscosity and density mismatches affect nebulization efficiency.
Tip: Match standard and sample matrices as closely as possible.

3. Instrument Calibration and Quality
Monochromator Efficiency:
Low-quality filters may cause spectral interference.
Solution: Use high-purity optical filters and validate them periodically.
Photodetector Sensitivity:
Aging photodetectors result in signal degradation.
Maintenance: Replace detectors regularly and keep calibration logs.
Environmental Interference:
Vibrations, air drafts, and temperature shifts destabilize flame conditions.
Best Practice: Place the instrument in a vibration-free, draft-free room with controlled temperature.
Optimizing Flame Photometry: Best Practices
Use deionized water for all dilutions and cleaning steps.
Filter samples to remove particulates before aspiration.
Adjust the fuel/oxidant ratio for a clean, blue, non-flickering flame.
Perform daily calibration using certified standard solutions.
Maintain a maintenance logbook for routine checks and part replacements.
Train all users in standard operating procedures and safety protocols.

Common Flame Photometer Errors and Solutions
| Issue | Likely Cause | Recommended Action |
|---|---|---|
| No Spark or Ignition Sound | Faulty power board or igniter | Check 5V output, replace transformer/pulse unit |
| Spark Sound but No Flame | Dirty ignition head | Clean ignition and combustion heads |
| Gas Smell but No Ignition | Incorrect gas concentration | Adjust gas valve (2–3 turns) |
| No Gas Smell, No Flame | Gas supply blockage | Check gas tank to burner pathway |
| Flame Unstable | Air/gas flow inconsistency | Adjust valve, check solenoid and thermocouple |
| Air Pressure Fluctuates | Blocked or damaged air filter | Clean or replace air filter |
| No Sample Suction | Blocked capillary or nebulizer | Unclog or replace sampling capillary |
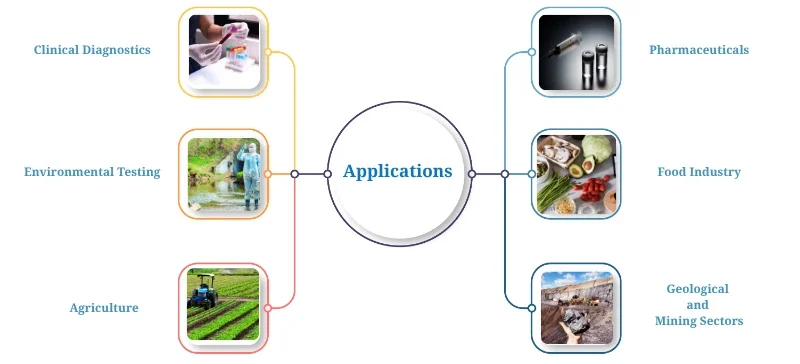
Applications of Flame Photometry
Clinical Diagnostics: Measuring sodium and potassium in blood and urine samples.
Environmental Testing: Detecting metal content in water, soil, and plant material.
Agriculture: Monitoring nutrient content in fertilizers and irrigation water.
Pharmaceuticals: Quality control for trace metal content.
Food Industry: Mineral analysis in beverages and processed food.
Geological and Mining Sectors: Elemental analysis of minerals and rocks
Conclusion
Flame photometry is a cost-effective, high-speed, and user-friendly analytical technique ideal for quantifying alkali and alkaline earth metals. To achieve accurate and reproducible results, users must carefully control flame conditions, maintain the instrument regularly, and adhere to standardized sample preparation methods. By implementing the optimization techniques and troubleshooting methods outlined in this guide, laboratories can significantly improve the performance and lifespan of their flame photometers—especially AELAB models.


















