Introduction
The bomb calorimeter is a critical scientific instrument widely used to measure the heat released during the combustion of a sample under controlled conditions. Accurate measurement of energy content in fuels, food, and various materials is vital for researchers, engineers, and industry professionals to enhance efficiency, safety, and product quality. This detailed guide covers the bomb calorimeter’s structure, how it works, and its broad range of applications, offering valuable knowledge for students and professionals alike.
Main Components of a Bomb Calorimeter
Bomb (Combustion Chamber): A strong, sealed container where the sample is combusted.
Ignition System: An electric ignition wire that initiates combustion.
Calorimeter Jacket: Contains water that absorbs heat from the bomb.
Temperature Sensor: Measures precise changes in water temperature.
Pressure Gauge: Monitors and ensures the bomb can safely contain combustion pressure.

Advantages and Disadvantages
Advantages
High Precision: Delivers accurate measurement of energy content.
Controlled Environment: Pure oxygen ensures complete combustion.
Versatile: Suitable for solid, liquid, and gaseous samples.
Reliable: Consistent results under standardized testing conditions.
Disadvantages
Complex Operation: Requires trained personnel.
Sample Limitations: Only combustible materials can be tested.
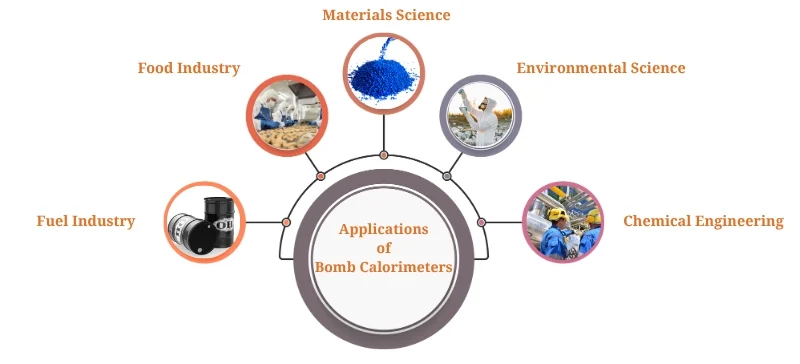
Applications of Bomb Calorimeter
These devices are extensively applied across multiple industries, including:
Fuel Industry: Measuring the calorific value of coal, petroleum, biofuels, and natural gas.
Food Industry: Determining the energy content of food products for nutritional labeling.
Materials Science: Studying combustion properties of polymers and other materials.
Environmental Science: Assessing energy from waste combustion.
Chemical Engineering: Optimizing fuel formulations and designing combustion reactors.
Bomb Calorimeter vs. Other Calorimetric Techniques
| Feature | Bomb Calorimeter | Coffee Cup Calorimeter | Differential Scanning Calorimetry (DSC) |
|---|---|---|---|
| Combustion Type | Constant volume, complete | Constant pressure | No combustion, heat flow analysis |
| Sample Type | Solids, liquids, gases | Mostly liquids | Various (polymers, solids) |
| Accuracy | Very high | Moderate | High for phase changes |
| Pressure | High oxygen pressure | Atmospheric | Controlled atmosphere |
| Typical Use | Energy content analysis | Heat of solution/reaction | Thermal properties and phase transitions |
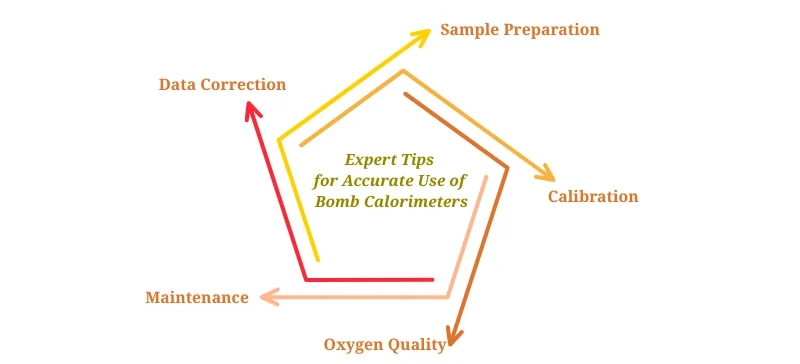
Expert Tips for Accurate Use of Bomb Calorimeter
Sample Preparation: Ensure the sample is dry and homogenous.
Calibration: Regularly calibrate with standard reference materials.
Oxygen Quality: Use high-purity oxygen to guarantee complete combustion.
Maintenance: Clean the bomb after each test to prevent residue buildup.
Data Correction: Adjust for heat losses and calorimeter heat capacity.
Conclusion
The bomb calorimeter remains the gold standard for precise measurement of combustion heat. Its durable construction and accurate operation make it indispensable in energy research, industrial quality control, and scientific studies. Whether analyzing fuels or food products, understanding its operation empowers you to obtain reliable energy data that can drive smarter decisions.
Interested in learning more about bomb calorimeters or upgrading your lab equipment? Get in touch with AELAB today for expert support, precision instruments, and advanced solutions tailored to your needs!