Introduction to Dissolution Testing and Dissolution Testers
Dissolution Testing System is crucial in the pharmaceutical industry as it measures the rate at which active pharmaceutical ingredients (APIs) are released from dosage forms. This system ensures drugs are absorbed into the bloodstream and become bioavailable. By simulating the human digestive system, the Dissolution Testing System allows manufacturers to evaluate how drugs dissolve in gastrointestinal fluids and how quickly they produce therapeutic effects.

Why is Dissolution Testing Crucial?
To be effective, a drug must dissolve properly in the stomach or intestines before entering the bloodstream. If the dissolution rate is too slow or too fast, the drug may not reach its intended target. This could reduce its effectiveness and present safety risks. Dissolution Testers provide insights into the drug’s release profile, ensuring that it dissolves at the right rate for predictable and consistent results in both formulation development and quality control.
The Role of Dissolution Testing System in the Pharmaceutical Industry
A Dissolution Testing System simulates the human digestive process by agitating the sample in a controlled medium. This method helps scientists understand how drugs release their active ingredients. The results provide valuable insights to optimize formulations, ensuring that drugs dissolve efficiently and consistently within the body.

How the Dissolution Testing System Works
The dissolution testing process involves several steps:
Preparation: Place the drug sample (tablet, capsule, etc.) into a vessel with simulated gastrointestinal fluid.
Agitation: Use rotating paddles or shafts to stir the sample, mimicking body movement to aid dissolution.
Temperature Control: Maintain a controlled temperature of 37.2°C, matching body temperature, for accurate results.
Sampling: At specific intervals, withdraw samples from the medium to measure the drug concentration. Calculate the dissolution rate based on these measurements.
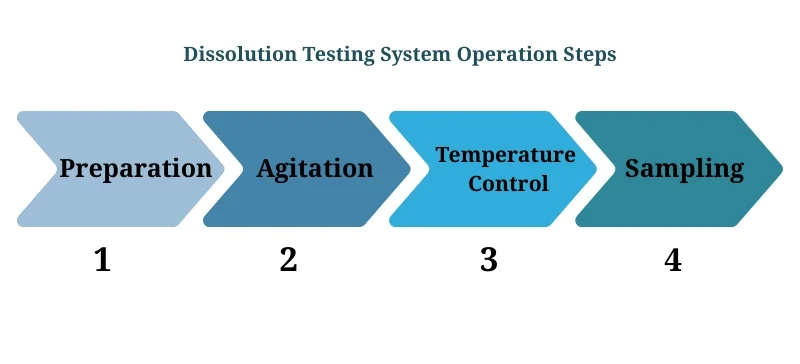
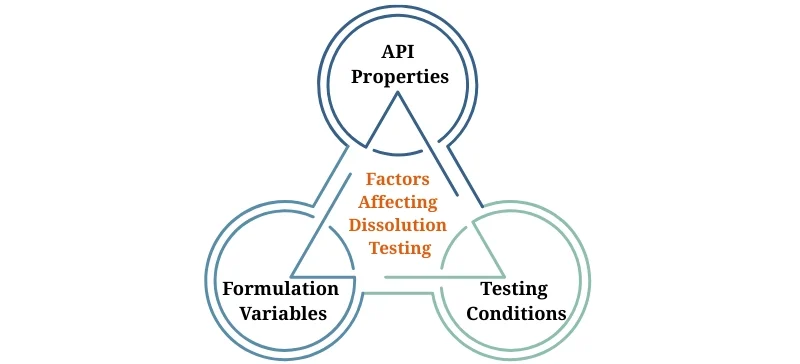
Factors Affecting Dissolution Testing
Several factors can influence dissolution, and Dissolution Testing Systems account for these variables:
API Properties: Solubility, particle size, and molecular structure affect the dissolution rate.
Formulation Variables: The choice of excipients, tablet hardness, and compression force influence how a drug dissolves.
Testing Conditions: The temperature, pH, and agitation rate must be optimized for accurate dissolution testing.
Dissolution Testing in Drug Development and Manufacturing
Early in drug development, Dissolution Testers help scientists optimize formulations. By understanding how changes in formulation affect the dissolution rate, they can ensure that drugs meet bioavailability requirements. Additionally, dissolution testing aids in establishing in vitro-in vivo correlations (IVIVCs), which predict how the drug will behave in the human body.
Once the drug moves into production, Dissolution Testers continue to ensure consistency. Regulatory bodies like the FDA and EMA require dissolution testing data before approving new drugs. The data also helps to monitor the drug’s quality post-market.
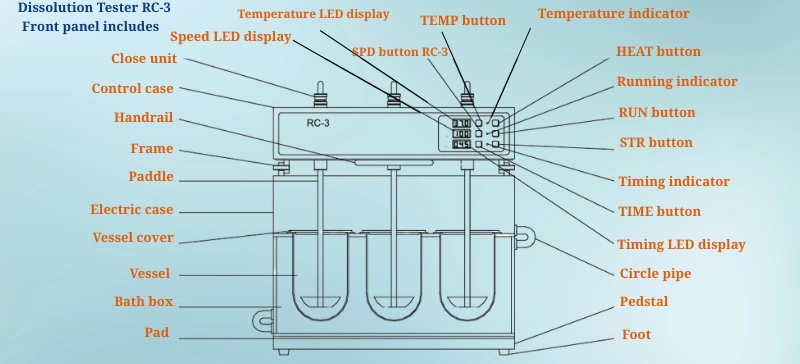
The Role of Automated Dissolution Testers
Automated Dissolution Testers, such as those offered by AELAB, provide accurate, reliable, and reproducible results. Automation reduces the risk of human error and allows for high-throughput testing, essential for large-scale production and stability studies. These systems help maintain consistent dissolution profiles across different batches, supporting both drug development and regulatory compliance.
Meeting Regulatory and Industry Standards
Dissolution Testers must meet rigorous standards set by global pharmacopeias, such as the United States Pharmacopeia (USP) and European Pharmacopoeia (EP). These standards ensure that dissolution testing is conducted uniformly, allowing for accurate and consistent results across various formulations and manufacturers. Adhering to these standards helps pharmaceutical companies gain market approval and maintain consumer trust.
Conclusion: The Essential Role of Dissolution Testers
Dissolution Testing Systems are essential for ensuring that drugs are safe, effective, and of high quality. They provide critical information about the drug’s release rate and behavior in the body. These systems play a pivotal role in drug development, quality control, and regulatory compliance. Whether for early-stage formulation optimization or large-scale manufacturing, dissolution testing remains a cornerstone of modern pharmaceutical science.
Call to Action
Improve the reliability and compliance of your pharmaceutical products by integrating automated Dissolution Testing Systems from AELAB. Equip your lab with state-of-the-art technology to ensure accurate, efficient, and compliant dissolution testing today!