Introduction
DSC vs TGA is one of the most common comparisons in the field of thermal analysis, especially across research laboratories and industrial environments. These two core techniques — Differential Scanning Calorimetry (DSC) and Thermogravimetric Analysis (TGA) — are widely used to evaluate material behavior under controlled temperature conditions.
But the key question remains:
What is the real difference between DSC and TGA — and which method is right for your material?
This comprehensive guide explains their differences, applications, data outputs, advantages, limitations, and even simultaneous analysis systems (STA) in a fully structured, SEO-optimized format suitable for modern AI-driven search systems.
What Is Thermal Analysis and Why DSC vs TGA Matter?
Thermal analysis refers to techniques used to evaluate how materials respond to temperature changes.
When discussing DSC vs TGA, thermal analysis helps answer:
At what temperature does a material melt? (DSC)
When does decomposition begin? (TGA)
Does the material lose mass during heating? (TGA)
What phase transitions occur? (DSC)
The two most essential techniques in any DSC vs TGA comparison are:
Differential Scanning Calorimetry (DSC)
Thermogravimetric Analysis (TGA)
What Is DSC? (Differential Scanning Calorimetry)
Scientific Definition of DSC
Differential Scanning Calorimetry (DSC) is a thermal analysis technique that measures the amount of heat absorbed or released by a sample during heating, cooling, or isothermal processes.
In DSC:
A sample and a reference are heated simultaneously.
The difference in heat flow between them is recorded.
Results are displayed as a Heat Flow vs Temperature curve.
What Does DSC Measure?
DSC provides critical insights into:
Glass transition temperature (Tg)
Melting temperature (Tm)
Crystallization behavior
Curing reactions
Specific heat capacity
Enthalpy changes
Oxidation Induction Time (OIT)
What Does a DSC Output Look Like?
The DSC thermogram includes:
Endothermic peaks → heat absorption (e.g., melting)
Exothermic peaks → heat release (e.g., crystallization or curing)
DSC is ideal for studying energy-related thermal events and phase transitions.
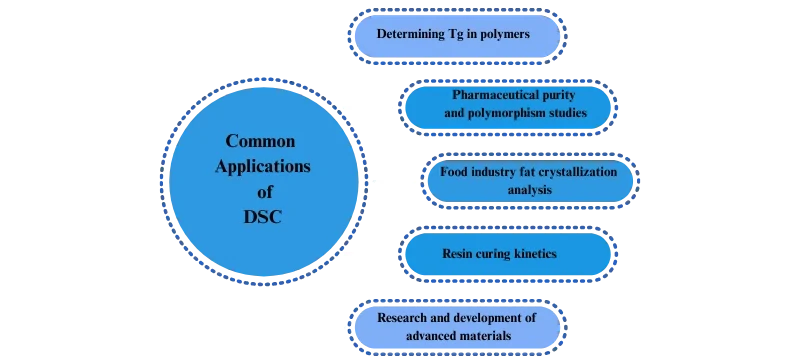
Common Applications of DSC
Determining Tg in polymers
Pharmaceutical purity and polymorphism studies
Food industry fat crystallization analysis
Resin curing kinetics
Research and development of advanced materials
What Is TGA? (Thermogravimetric Analysis)
Scientific Definition of TGA
Thermogravimetric Analysis (TGA) is a technique that measures changes in a material’s mass as a function of temperature or time under a controlled atmosphere.
In TGA:
The sample is placed on a high-precision microbalance.
Temperature increases according to a programmed heating profile.
Weight changes are continuously recorded.
What Does TGA Measure?
TGA provides information about:
Moisture content
Volatile components
Thermal decomposition temperature
Oxidation behavior
Filler or ash content
Thermal stability
Material composition
What Does a TGA Output Look Like?
TGA results include:
TG Curve (Mass vs Temperature)
DTG Curve (Derivative Mass Loss)
Each weight-loss step may indicate:
Evaporation
Thermal decomposition
Combustion
Release of volatile compounds
What Is the Main Difference Between DSC vs TGA?
| Feature | DSC | TGA |
|---|---|---|
| Measurement | Heat flow (energy change) | Mass (weight change) |
| Key Question | How much energy changes? | How much mass changes? |
| Focus | Phase transitions | Stability & decomposition |
| Output | Heat flow curve | TG & DTG curves |
| Best For | Tg, melting, crystallization | Moisture, composition, degradation |
When Should You Use DSC?
Choose DSC if you need to:
Determine glass transition temperature (Tg)
Analyze melting behavior
Study curing reactions
Calculate crystallinity
Investigate phase transitions
Measure oxidation induction time
→ DSC is the appropriate method.
When Should You Use TGA?
Choose TGA if your objective is to:
Measure moisture content
Determine filler percentage (e.g., CaCO₃, glass fiber)
Study thermal degradation
Analyze polymer decomposition
Calculate ash or char yield
Evaluate oxidation resistance
→ TGA is the correct choice.
Industry-Based DSC vs TGA Comparison
Polymers & Plastics in DSC vs TGA
Tg and melting → DSC
Filler content and degradation → TGA
Pharmaceuticals in DSC vs TGA
Polymorphism and purity → DSC
Water of hydration → TGA
Batteries & Energy Materials in DSC vs TGA
Separator melting → DSC
Binder content → TGA
AELABPrivate: AELAB Differential Scanning Calorimeter AE-DSC600 – Precision Thermal Analysis
Can DSC Measure Decomposition Temperature?
Not reliably on its own.
DSC does not measure mass loss. For accurate confirmation of thermal decomposition, TGA is required.
Can TGA Measure Melting Point?
No.
If melting occurs without mass change, TGA cannot detect it.
For accurate melting temperature measurement → DSC is necessary.
Advantages and Limitations
Advantages of TGA
Excellent for composition analysis
High-temperature capability (up to 1600°C in advanced systems)
Ideal for thermal degradation studies
Multi-atmosphere flexibility
Limitations of TGA
Cannot detect phase transitions without mass change
Does not measure enthalpy
Advantages of DSC
Detects subtle thermal transitions
Measures enthalpy changes
Essential for Tg analysis
IIdeal for polymer and pharmaceutical R&D
Limitations of DSC
Cannot measure mass loss
Limited decomposition insight when used alone
How to Choose Between DSC vs TGA
Your decision when choosing between DSC vs TGA depends on three key factors:
Material type (polymer, metal, ceramic, pharmaceutical, composite)
Research objective (energy profiling vs stability assessment)
Sample behavior under heat
Conclusion
Understanding DSC vs TGA is essential for selecting the correct thermal analysis technique and ensuring reliable material characterization. While DSC provides critical insights into heat flow, phase transitions, and thermodynamic behavior, TGA focuses on mass changes, decomposition patterns, and thermal stability.
Neither technique replaces the other — instead, they complement each other. Choosing the right method depends on your material type, research objective, and required data accuracy.
For laboratories seeking comprehensive thermal insight, combining DSC and TGA offers a more complete and confident understanding of material performance under heat. In modern analytical environments, this integrated approach represents the most effective strategy for advanced thermal characterization.