Introduction
In clinical laboratories, the accuracy of diagnostic results hinges on the performance of automated biochemistry analyzers, which are used to measure concentrations of substances such as glucose, cholesterol, enzymes, and proteins in biological samples. Calibration is a critical process that ensures these analyzers provide precise and reliable data, which is essential for accurate diagnosis, treatment planning, and patient monitoring. This article explores what effective calibration is, why it matters, and how it can be optimized to meet the highest clinical standards.
What is Calibration in Biochemistry Analyzers?
Calibration refers to the process of configuring a biochemistry analyzer to produce results that align with known standards or reference values. It involves measuring a calibration standard or solution with a known concentration and adjusting the analyzer’s output to match the true value. This helps correct deviations caused by instrument drift, reagent variability, or environmental factors.
Types of Calibration:
Initial Calibration: Establishes the baseline response when a new analyzer or test is initiated.
Recalibration: Performed after maintenance or when performance drifts.
Calibration Verification: Confirms the accuracy of a completed calibration.
Two-Point Calibration: Used for linear assays; fast and simple.
Multi-Point Calibration: Ideal for non-linear assays requiring complex curves.
Batch Calibration: Applied in semi-automated or batch-mode analyzers.
Continuous Calibration: Real-time calibration in high-throughput analyzers.
Why is Effective Calibration Important?
Ensuring Accuracy and Reliability
Regular calibration ensures that test results reflect actual analyte concentrations, supporting accurate diagnoses and effective clinical decisions using AELAB biochemistry analyzers.
Compensating for Instrument Drift
Factors like mechanical wear and temperature changes can affect analyzer performance over time. Calibration resets the instrument’s measurement baseline to maintain consistency.
Consistency Across Reagent Batches
Calibration compensates for variability between reagent lots, ensuring reliable results regardless of batch changes.
Compliance with Regulatory Standards
Standards from CLIA, CAP, and ISO 15189 mandate calibration and documentation to verify instrument accuracy. Non-compliance risks accreditation and patient safety.
Foundation for Quality Control
Effective calibration supports accurate quality control, ensuring QC results are trustworthy and that erroneous patient results are avoided.
Protecting Patient Safety
Incorrect calibration can lead to misdiagnosis or inappropriate treatment. Proper calibration protects patients from these risks.
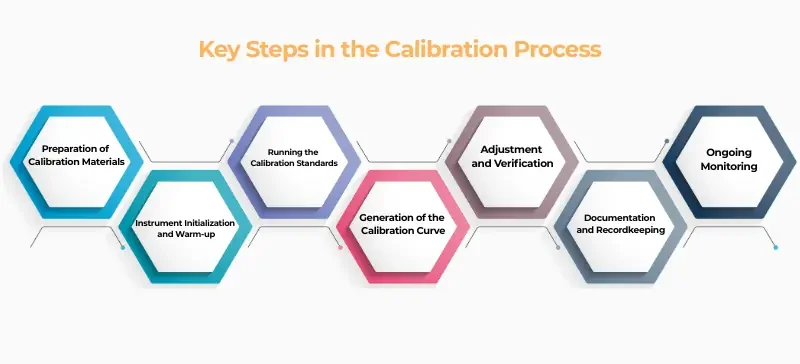
Key Steps in the Calibration Process
1. Preparation of Calibration Materials
Use traceable, certified calibration standards stored properly. Match calibrators to assay requirements and instrument specs.
2. Instrument Initialization and Warm-up
Allow the analyzer to stabilize before calibration. Skipping this step may lead to fluctuating results.
3. Running the Calibration Standards
Process standards as you would patient samples. Use multiple standards across the assay’s range for accuracy.
4. Generation of the Calibration Curve
The analyzer creates a curve (linear, polynomial, etc.) linking signal intensity to analyte concentration.
5. Adjustment and Verification
Adjust analyzer settings based on calibration data. Validate with QC samples to confirm reliability.
6. Documentation and Recordkeeping
Log every calibration event, including date, materials, and technician details, to ensure traceability and audit readiness.
7. Ongoing Monitoring
Monitor analyzer performance daily. Recalibrate based on QC trends, reagent lot changes, or system alerts.
Common Challenges in Calibration
Reagent and Calibrator Instability
Degradation due to poor storage can skew calibration results. Follow manufacturer guidelines for handling and storage.
Instrument Drift and Mechanical Wear
Aging components can affect accuracy. Regular maintenance and recalibration are essential.
Incorrect Calibration Frequency
Over- or under-calibration affects efficiency and reliability. Use QC trends to determine optimal timing.
Human Error in Setup and Execution
Mistakes in preparing standards or data entry can compromise calibration. Ensure training and SOPs are in place.
Interference and Contamination
Carryover, hemolysis, or lipemia can distort calibration. Maintain clean sample paths and check for interference.
Software or Algorithm Limitations
Poor curve fitting or software bugs may cause errors. Keep software updated and verify calibration models.
Reagent Lot-to-Lot Variability
Differences between reagent batches require recalibration. Perform parallel testing to confirm consistency.

Best Practices for Effective Calibration of Biochemistry Analyzers
| Best Practice | Explanation | Benefit |
|---|---|---|
| Use Certified Calibrators | Traceable to international standards | Ensures consistent, reliable results |
| Follow Manufacturer’s Instructions | Adhere to recommended procedures | Prevents procedural errors |
| Verify Calibration with QC Samples | Run quality control samples post-calibration | Confirms calibration success |
| Document Every Calibration Event | Record all calibration parameters | Enables traceability and compliance |
| Maintain Clean Sample Paths | Prevent contamination or carryover | Ensures data integrity |
| Monitor for Drift or Inconsistency | Analyze QC trends | Allows early error detection |
| Schedule Regular Recalibration | Use system alerts and performance trends | Ensures long-term analyzer accuracy |
| Train Staff Continuously | Update and reinforce technician knowledge | Reduces human error |
| Use the Right Calibration Type | Match calibration to assay complexity | Enhances result precision |
| Inspect Reagents and Calibrators | Check storage and expiration | Prevents using degraded materials |
Calibration vs. Quality Control
Calibration sets the baseline; quality control ensures that baseline holds over time. Both are essential for accuracy.
| Feature | Calibration | Quality Control |
| Purpose | Adjust accuracy | Verify ongoing performance |
| Frequency | Scheduled or as needed | Routine (daily or per batch) |
| Materials Used | Reference standards | Control samples |
Conclusion
Calibration of biochemistry analyzers is a foundational process that safeguards the accuracy and reliability of laboratory testing. Through systematic calibration practices, the use of certified standards, and adherence to regulatory requirements, laboratories using AELAB analyzers can ensure optimal performance, protect patient safety, and maintain accreditation.
Ready to Optimize Your Lab’s Accuracy?
Review your calibration protocols or contact our expert team for a customized evaluation of your AELAB analyzer setup.