Comparing Gas and Liquid Chromatography: Techniques, Differences, and Applications
Chromatography is a crucial separation technique widely used in analytical chemistry to separate and identify compounds in a mixture. Two of the most prominent forms of chromatography are Gas Chromatography (GC) and High-Performance Liquid Chromatography (HPLC). While both techniques are highly effective for compound separation and analysis, they differ significantly in terms of the mobile phase, operational principles, and ideal applications. This article provides a comparative analysis of GC and HPLC, discussing their respective strengths, limitations, and typical use cases across various industries.

Learn More:Window Tint Meters: Applications in Various Industries
Fundamental Differences
Mobile Phase
The primary distinction between GC and HPLC lies in the type of mobile phase used. Gas Chromatography relies on a gas (often helium or nitrogen) as the mobile phase, which is suitable for volatile and thermally stable compounds. In contrast, High-Performance Liquid Chromatography uses a liquid mobile phase, typically consisting of mixtures of solvents with compatible polarities. This makes HPLC suitable for analyzing non-volatile, polar, or larger molecular compounds that cannot withstand high temperatures.
Operating Temperature
GC typically operates at elevated temperatures, essential for maintaining the volatility of the compounds under analysis. The temperature conditions can be controlled in an isothermal or temperature-programmed manner, depending on the specific application. On the other hand, HPLC generally operates at ambient temperatures, as many compounds, especially biomolecules and large polymers, may degrade at higher temperatures.
Nature of Compounds
The nature of the compounds being analyzed plays a crucial role in the choice of technique. GC is particularly effective for volatile compounds with lower molecular weights, typically ranging from a few hundred Daltons. It separates compounds based on differences in volatility and is particularly suited for analyzing gases, oils, fragrances, and environmental pollutants. HPLC, on the other hand, is optimal for separating non-volatile, highly polar, or large biomolecules, including proteins, peptides, pharmaceuticals, and food additives.
Column Dimensions and Packings
In GC, longer and narrower columns accommodate gases with lower viscosity, allowing them to travel through the stationary phase with minimal resistance. This design enables higher resolution separations, especially for volatile compounds. On the other hand, HPLC uses shorter and wider columns because liquids have higher viscosity, which increases column back pressure. The packing materials in HPLC columns are selected based on the polarity and molecular size of the compounds, whereas in GC, the volatility of the compounds determines the separation process.
Figure 1 below demonstrates how a High-Performance Liquid Chromatography (HPLC) system operates. The diagram highlights key components such as the mobile phase, pump, injector, column, detector, and data acquisition system, explaining how samples undergo injection, separation, and analysis.
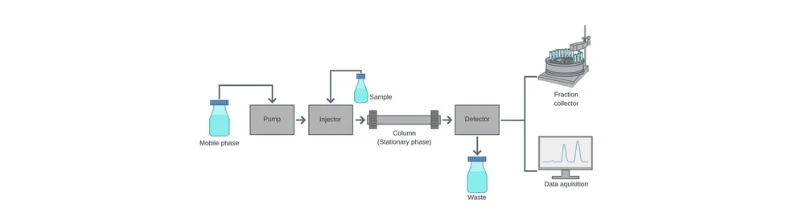
Figure 1: Schematic representation of the HPLC process, detailing the injection, separation, and detection stages.
Detection Principles
Both techniques offer highly sensitive detection methods, but the principles differ. HPLC typically employs non-destructive detectors, such as UV absorbance, Refractive Index (RI), and conductivity detectors, which do not alter the chemical structure of the compounds during detection. GC, however, uses destructive detectors, such as Flame Ionization Detectors (FID), Nitrogen-Phosphorus Detectors (NPD), and Mass Spectrometry (MS), which break down the compounds during the detection process. Mass spectrometric detection is used in both GC and HPLC for ultra-sensitive analysis and structural elucidation.Figure 2 below demonstrates the operational workflow of a Gas Chromatography (GC) system, including the injection of the sample, separation within the coiled column, and detection of compounds based on their volatility and solubility.
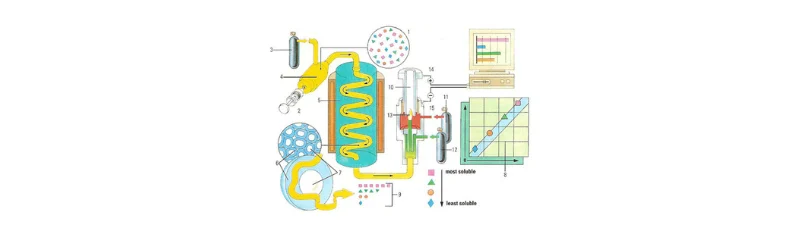
Figure 2: Diagram of a Gas Chromatography system, illustrating the sample injection, separation in the coiled column, and detection of compounds.
Operational Complexity and Cost
GC is generally considered easier to operate and requires lower initial investment compared to HPLC systems. The operation of GC is less complex, as it primarily involves controlling temperature and gas flow. However, the need for volatile samples can necessitate extensive sample preparation, such as derivatization. HPLC, on the other hand, is more complex due to the variety of mobile and stationary phases that can be used, as well as the high-pressure pumps needed for liquid chromatography. Consequently, HPLC systems tend to have higher operational and maintenance costs. However, HPLC’s ability to handle a wider range of compounds offsets these costs.
Sample Requirements
GC typically requires smaller sample volumes than HPLC, due to the high sensitivity of GC detectors. The small sample sizes are particularly advantageous when analyzing scarce or valuable samples. On the other hand, HPLC may require larger sample volumes, especially when the sample needs pre-column treatment or complex analysis. The higher sample volume requirement for HPLC can be mitigated by its greater versatility in analyzing a broader spectrum of compounds.
Advantages and Limitations
Each chromatography technique has distinct advantages and limitations based on the type of sample and the application at hand.
-
GC Advantages
- Excellent for analyzing volatile and thermally stable compounds.
- Higher sensitivity for small sample volumes.
- Lower operational costs, especially when analyzing simpler compounds.
- Widely used in environmental monitoring (e.g., air pollutants) and flavor analysis.
-
GC Limitations
- Can only be used for volatile compounds.
- Requires sample derivatization for non-volatile compounds, which can add to the complexity of analysis.
-
HPLC Advantages
- Can analyze a wide range of compounds, including non-volatile, polar, and large biomolecules.
- Higher separation efficiency and broader sample adaptability.
- Ideal for analyzing biopharmaceuticals, food safety, and complex biological samples.
-
HPLC Limitations
- Requires larger sample volumes.
- Higher initial and maintenance costs, particularly when using high-end detectors like Mass Spectrometry (MS).
Industry Applications
Gas Chromatography is used in environmental science, petroleum refining, pharmaceuticals, food, and forensic analysis. It detects volatile organic compounds (VOCs) and monitors air quality, as well as analyzes food flavors and fragrances.
High-Performance Liquid Chromatography is essential in biopharmaceuticals, food safety, and pharmaceutical analysis. It analyzes complex, non-volatile, or thermally sensitive molecules like proteins, peptides, and DNA/RNA. It is crucial for clinical diagnostics and genomic research.
Advanced Chromatography Solutions by AELab
AELab offers a diverse range of advanced chromatography instruments, specializing in both Gas Chromatography (GC) and High-Performance Liquid Chromatography (HPLC) systems. These include state-of-the-art models such as the Gas Chromatography GC1120, the Gas Chromatograph Mass Spectrometer GC-MS 3200, and the, which are mentioned as examples. AELab also provides a variety of other models with unique features and applications, catering to specific analytical needs across industries such as food safety, environmental monitoring, and chemical analysis. These instruments highlight AELab’s commitment to providing cutting-edge solutions for separating, identifying, and quantifying compounds in complex mixtures.

All-in-One Smart Isocratic System High Performance Liquid Chromatography LC-80
Conclusion
In summary, both Gas Chromatography (GC) and High-Performance Liquid Chromatography (HPLC) are powerful analytical techniques that offer unique advantages depending on the nature of the sample and the specific requirements of the analysis. GC is best suited for analyzing volatile, thermally stable compounds, and is more cost-effective for routine analyses. In contrast, HPLC provides greater versatility and is ideal for analyzing a broader range of compounds, including biomolecules and non-volatile substances.
Choosing the appropriate chromatography technique involves considering factors such as sample properties, analysis goals, cost considerations, and required sensitivity. By understanding the strengths and limitations of GC and HPLC, researchers and industry professionals can make informed decisions to ensure the accuracy, efficiency, and reliability of their analyses across diverse scientific and industrial domains.
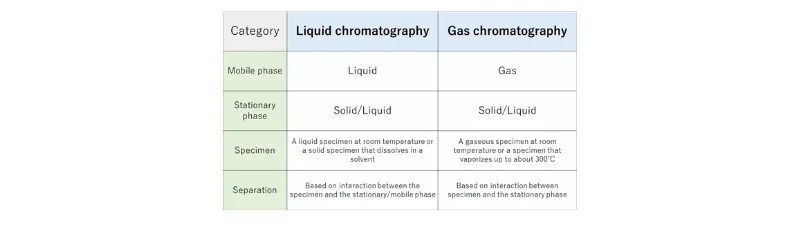 Figure 3: Comparative summary of the fundamental differences between HPLC and GC.
Figure 3: Comparative summary of the fundamental differences between HPLC and GC.