What Are the Water Quality Needs of HPLC?
Water is not merely a convenient solvent in High-Performance Liquid Chromatography (HPLC); it is a critical analytical reagent. In many chromatographic methods—especially gradient elution, trace-level analysis, and UV or MS detection—the quality of water directly determines baseline stability, sensitivity, reproducibility, and column lifetime.
Even trace amounts of ionic, organic, particulate, or microbial contamination in water can compromise results. This is why understanding the exact water quality requirements for HPLC is essential for any laboratory seeking reliable, publication-quality data.
This guide explains:
What “HPLC-grade water” really means
Why distilled or deionized water often fails
The key quality parameters that matter most
How water contamination appears in chromatograms
Best practices for producing, handling, and storing HPLC water
Why Water Quality Matters in HPLC
Modern HPLC systems are designed to detect extremely small signals. While this improves detection limits, it also makes the technique highly sensitive to impurities introduced through the mobile phase.
oor water quality commonly leads to:
Ghost peaks and unknown peaks
Baseline noise or drift (especially in gradient runs)
Retention time variability
Poor peak shape and resolution
Increasing backpressure
Column fouling and shortened column life
Higher maintenance costs and instrument downtime
In short, if the water is not clean enough, the chromatogram will reveal it.
What Water Should Be Used for HPLC?
The Standard Answer For most HPLC applications, the gold standard is Type I ultrapure water.
Typical Specifications for HPLC Water
Resistivity: 18.2 MΩ·cm at 25 °C
Total Organic Carbon (TOC): ≤ 5 ppb
UV absorbance: Extremely low (especially at 214 and 254 nm)
Particles: Removed by 0.22 µm final filtration
Microbial contamination: Minimal and well controlled
Important: High resistivity alone does not guarantee suitability for HPLC. Water can show excellent resistivity while still containing enough organic contamination to ruin a gradient method.
Key Water Quality Parameters for HPLC
1. Resistivity (Ionic Purity)
Resistivity reflects how effectively ions have been removed. Ionic contamination can:
Alter retention and selectivity of ionizable compounds
Introduce UV-active species (e.g., nitrates)
Reduce method reproducibility
Target value: ~18.2 MΩ·cm
2. Total Organic Carbon (TOC)
TOC is often the most critical parameter for modern HPLC.
Trace organic compounds can:
Accumulate on the column
Elute later as ghost peaks
Increase baseline noise and drift
Change selectivity over time
Target value: ≤ 5 ppb, especially important for gradient elution and trace analysis.
3. UV Absorbance
For UV-based detection, water must have minimal absorbance at analytical wavelengths. Low absorbance—especially below 220 nm—is a practical indicator of very low organic contamination.
4. Particulate Content
Particles can:
Clog column inlet frits
Increase system backpressure
Damage pump seals and injector valves
Shorten column lifetime
Best practice: 0.22 µm filtration of water and mobile phases.
5. Microbial Control
Ultrapure water can still support microbial growth if stored or if purification systems are poorly maintained. Microorganisms:
Release organic byproducts (raising TOC)
Form biofilms
Reduce reproducibility and system reliability

Is the pH of Ultrapure Water Important?
Not in the conventional sense.
Ultrapure water contains very few ions, so pH electrode readings are unstable and unreliable. Additionally, ultrapure water rapidly absorbs CO₂ from air, forming carbonic acid and changing apparent pH.
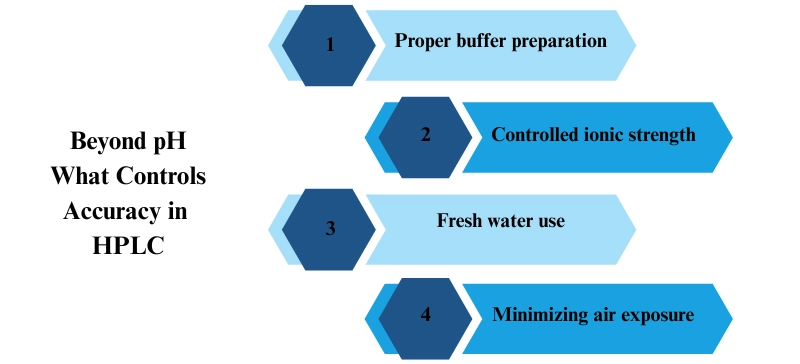
Instead of focusing on the pH of pure water, laboratories should focus on:
Proper buffer preparation
Controlled ionic strength
Fresh water use
Minimizing air exposure
How Type I Water for HPLC Is Produced
Reliable HPLC water cannot be produced by a single purification step. High-quality systems use a multi-barrier purification approach, typically including:
Pretreatment → Reverse Osmosis → Deionization → UV Oxidation (185 nm) → UV Disinfection (254 nm) → Ultrafiltration → 0.22 µm Final Filter
This architecture ensures removal of ions, organics, microorganisms, particles, and endotoxins.
In-House (Point-of-Use) Ultrapure Water Pros
Fresh water with consistent quality
Full control of TOC and resistivity
Lower cost per liter for medium-to-high usage
Supports multiple analytical techniques
Cons
Requires regular maintenance and monitoring
For laboratories running gradients daily or performing LC-MS or trace analysis, point-of-use Type I water is usually the superior choice.
Best Practices for Handling and Storage of HPLC Water
Ultrapure water becomes contaminated quickly. The purer it is, the more aggressively it absorbs contaminants.
Recommended practices:
Use freshly produced water whenever possible
Collect water directly into clean, dedicated containers
Use glass or fluoropolymer containers reserved for HPLC solvents
Minimize air contact
Label collection time and avoid long storage
Avoid:
Long-term storage in plastic wash bottles
Topping off old mobile phases
Leaving containers open
Storing mobile phases longer than necessary
How Water Contamination Appears in Chromatograms
| Contaminant Type | Typical Effect |
|---|---|
| Organic compounds | Ghost peaks, baseline drift, tailing |
| Ions | Retention time shifts, selectivity changes |
| Particles | Increased backpressure, clogged frits |
| Microbial growth | Rising TOC, poor reproducibility |
| Dissolved gases | Noise, bubbles, unstable baseline |
Quick Troubleshooting: Water-First Checklist
Ghost peaks in a blank gradient
Prepare fresh mobile phase with newly produced ultrapure water
Check TOC polishing stage and UV lamp performance
Increasing backpressure
Filter mobile phase through 0.22 µm filters
Inspect inline filters and column frits
Consider microbial contamination
Retention time drift
Confirm stable resistivity
Avoid topping off solvents
Clean column if organic fouling is suspected
Conclusion
The water quality needs of HPLC extend far beyond “clean water.” For reliable, reproducible chromatography—especially in gradient methods and sensitive detection—water must be treated as a critical component of the analytical method.
Using properly produced and well-handled Type I ultrapure water with:
High resistivity (~18.2 MΩ·cm)
Ultra-low TOC (≤ 5 ppb)
Low UV absorbance
Effective particle and microbial control
results in quieter baselines, more robust methods, longer column life, and lower operating costs.