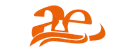Introduction
Ensuring the safety, efficacy, and quality of pharmaceutical products is a fundamental requirement in modern healthcare. Moreover, as drug formulations become increasingly complex and global regulations tighten (USP <232>/<233>, ICH Q3D, EMA), advanced analytical techniques are more critical than ever. In particular, ICP-MS in pharmaceuticals has established itself as the gold standard for detecting and quantifying elemental impurities at ultra-trace levels. Consequently, pharmaceutical companies rely on ICP-MS to maintain compliance and safeguard patient health.
What is ICP-MS and How Does It Work?
ICP-MS (Inductively Coupled Plasma Mass Spectrometry) is an advanced analytical technique that combines two powerful technologies:
Inductively Coupled Plasma (ICP): Generates a plasma torch at >6,000 °C to ionize the sample.
Mass Spectrometry (MS): Separates and quantifies ions based on their mass-to-charge ratio (m/z).
This unique combination allows ICP-MS to:
Detect elements at parts-per-trillion (ppt) levels.
Analyze up to 70 elements simultaneously in one run.
Perform isotopic and multi-element analysis with unmatched accuracy.
Key Applications of ICP-MS in Pharmaceuticals
1. Elemental Impurity Testing in APIs and Finished Products
For example, ICP-MS in pharmaceuticals ensures ultra-trace detection of toxic metals such as arsenic (As), cadmium (Cd), lead (Pb), and mercury (Hg), helping products meet strict regulatory standards.
2. Excipients and Formulation Testing
Additionally, even inactive excipients can introduce impurities during manufacturing. ICP-MS provides precise analysis to confirm excipient purity and safety.
3. Monitoring Metal Catalysts in Drug Synthesis
Many drugs are synthesized using metal-based catalysts like platinum, palladium, or rhodium. Therefore, ICP-MS verifies that residual metals remain below regulatory thresholds.
4. Stability Studies & Shelf-Life Testing
Over time, trace metals may leach into pharmaceuticals. ICP-MS helps manufacturers monitor degradation products and assess drug stability, thus ensuring long-term safety.

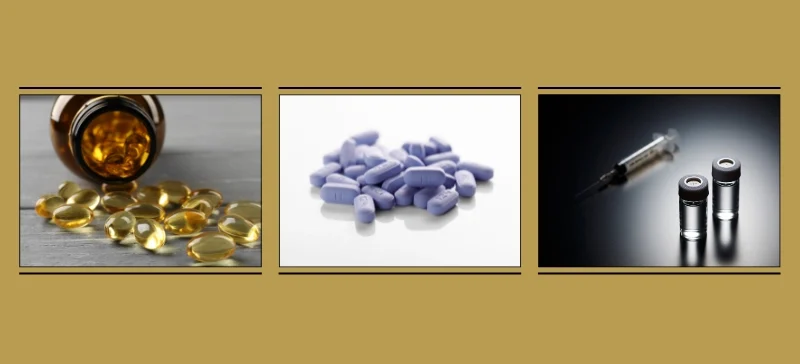
Advantages of ICP-MS in Pharmaceuticals Quality Control
- Ultra-Sensitivity: Detects contaminants at ppt levels.
- Multi-Element Capability: Measures dozens of elements in a single run.
- High Throughput: Suitable for labs handling large sample volumes.
- Isotopic Analysis: Valuable for research and drug development.
- Regulatory Compliance: Accepted globally by FDA, EMA, USP, and ICH.
- Broad Dynamic Range: Effective for both trace and higher concentrations.
Moreover, the versatility and speed of ICP-MS in pharmaceuticals make it indispensable for modern quality control.
Challenges of ICP-MS and Solutions
Matrix Interference: Complex drug formulations may distort results.
Solution: Use collision/reaction cells and internal standards.
Sample Preparation Complexity: Tablets, gels, and biologics require digestion.
Solution: Employ microwave-assisted digestion and automated prep systems.
Risk of Contamination: Even trace contamination can alter results.
Solution: Use cleanrooms, ultra-pure reagents, and blank testing.
Calibration Drift & Sensitivity Issues: May cause inaccurate results. Solution: Frequent calibration using certified reference materials.
High Capital and Operational Costs: ICP-MS instruments are expensive. Solution: Optimize sample throughput and partner with reliable providers.
Need for Skilled Personnel: Operation and data interpretation are complex.
Solution: Continuous staff training and advanced software.
ICP-MS vs Other Techniques
| Technique | Sensitivity | Multi-Element Detection | Speed | Regulatory Acceptance |
|---|---|---|---|---|
| ICP-MS | Excellent (ppt) | Yes | High | Global Standard |
| ICP-OES | Moderate (ppb) | Yes | High | Limited |
| AAS | Low | No | Slow | Less Preferred |
ICP-MS clearly stands out as the most sensitive, versatile, and compliant method for pharmaceutical impurity testing.
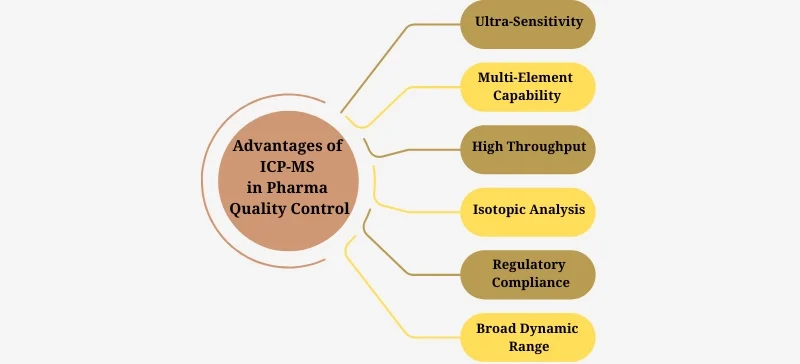
Expert Recommendations for Pharma Companies
Validate all ICP-MS methods according to ICH Q2 guidelines.
Use internal standards for accurate quantification.
Invest in advanced sample preparation systems for efficiency.
Partner with trusted ICP-MS solution providers.
Train QC analysts in both operation and data interpretation.
Conclusion: The Essential Role of ICP-MS in Pharmaceuticals for Drug Safety and Quality
ICP-MS in pharmaceuticals has transformed quality control by offering unmatched sensitivity, high throughput, and strict compliance with international standards. Furthermore, from monitoring elemental impurities in APIs and excipients to evaluating stability in biologics and supplements, ICP-MS protects patient safety and strengthens regulatory confidence.
As regulations continue to tighten, ICP-MS in pharmaceuticals is no longer optional; it is a crucial component for ensuring drug safety, efficacy, and global market compliance. Therefore, pharmaceutical companies aiming for accuracy, compliance, and reliability should integrate ICP-MS into their quality control workflows.
👉 Pharmaceutical companies seeking reliability and compliance should consider integrating ICP-MS solutions from AELAB into their quality control workflows.