Applications of ICP-OES in Monitoring Water Quality: Focus on Toxic Metals and Contaminants
Introduction
Inductively Coupled Plasma Optical Emission Spectrometry (ICP-OES) is a highly effective technique used for monitoring water quality, particularly in detecting toxic metals and contaminants. This instrument offers unparalleled sensitivity and precision, making it essential for the analysis of trace metals in water. This instrument can detect harmful elements such as arsenic, mercury, cadmium, and lead even at extremely low concentrations, which is crucial for ensuring water safety and regulatory compliance. Due to its ability to handle complex sample matrices, ICP-OES is widely used in environmental monitoring. This article explores the applications of this advanced instrument in water quality analysis, focusing on its role in detecting toxic metals and contaminants.
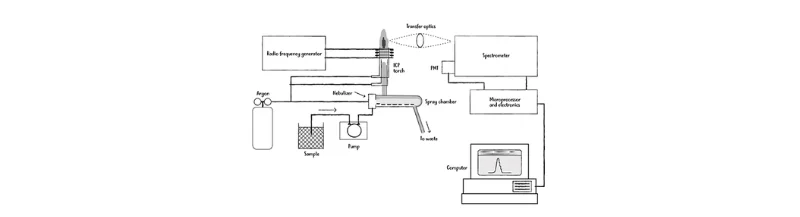
Figure 1: ICP-OES System Overview
Illustration of the main components of an ICP-OES system, including the nebulizer, spray chamber, ICP torch, and spectrometer, used for detecting toxic metals in water samples.
ICP-OES in Water Quality Monitoring
Evaluation of ICP-OES for Detecting Toxic Metals in Water Samples
ICP-OES is commonly used to detect toxic metals such as arsenic, mercury, cadmium, and lead in water. Even at trace concentrations, these metals pose significant risks to human health and ecosystems. This instrument provides the high sensitivity and accuracy necessary for regulatory compliance. The technique’s ability to detect metals in the nanogram-per-liter (ng/L) range ensures the quantification of even low-level contaminants, making it crucial for monitoring drinking water quality.
Role of ICP-OES in Routine Water Quality Testing for Environmental Compliance
Routine water testing is vital to ensure safe drinking water. ICP-OES is frequently used in monitoring programs to meet environmental regulations like the Safe Drinking Water Act (SDWA) in the U.S. and similar standards worldwide. Its ability to analyze multiple elements simultaneously makes it an efficient tool for testing large volumes of water for a wide range of contaminants. The technique can also analyze both major ions and trace elements, providing a comprehensive assessment of water quality.

learn more:Gas Chromatography vs Liquid Chromatography: Comparative Analysis and Use Cases
Identification of Toxic Metals in Water
Key Toxic Metals in Water: Arsenic, Mercury, and Cadmium Detection Challenges
Arsenic, mercury, and cadmium are some of the most dangerous contaminants in water. These metals can come from natural sources or industrial activities, making their detection critical for public health. ICP-OES is effective for detecting these metals, but challenges remain. For example, mercury requires specific protocols due to its volatility, and arsenic detection can be affected by interference from other substances in the sample. Addressing these challenges is key to accurate analysis of these critical contaminants.
Impact of Sample Matrix on the Detection of Trace Metals in Water Using ICP-OES
The sample matrix can significantly affect ICP-OES measurements. Factors like salinity, pH, and the presence of organic matter or suspended particles can cause interferences or signal suppression. These effects must be addressed during sample preparation and analysis to ensure reliable results. Common strategies to minimize these issues include sample digestion, matrix-matched calibration, and internal standardization.
ICP-OES vs. Other Analytical Techniques for Water Quality Monitoring
Though this instrument is highly effective for routine water quality analysis, other methods may be preferred for specific applications. Techniques like Graphite Furnace Atomic Absorption Spectroscopy (GF-AAS) and Atomic Fluorescence Spectroscopy (AFS) are often used for detecting metals like mercury and arsenic at even lower concentrations. However, This instrument offers significant advantages in multi-element analysis, speed, and lower detection limits for a broad range of metals.
Challenges in Toxic Metal Detection
Interferences in ICP-OES Analysis
ICP-OES can be affected by spectral and chemical interferences. Overlapping emission lines, especially for metals with similar wavelengths, complicate the quantification of target analytes. Matrix interferences, such as chloride ions, can alter plasma conditions and reduce sensitivity for certain metals. Advanced techniques like collision/reaction cell (CRC) technology can help mitigate these issues, improving it’s performance for difficult-to-analyze elements like arsenic and selenium.
Detection Limits and Regulatory Compliance
Achieving low detection limits (MDLs) is essential for regulatory compliance in water quality monitoring. For example, the U.S. EPA sets strict limits for metals like arsenic (10 ppb) and mercury (2 ppb) in drinking water. This instrument can achieve these limits, though advanced techniques like CRC ICP-MS may further improve sensitivity for some metals. Nonetheless, ICP-OES remains a widely accepted and practical choice for routine compliance testing.
Introduction of AELAB ICP-OES Products for Water Quality Monitoring
AELAB offers advanced spectrometers designed for water quality monitoring, such as the ICP-AES Spectrometer 4820/4850 and ICP-OES Spectrometer 6500. These instruments are optimized for fast, accurate analysis of trace metals in water, including arsenic, mercury, cadmium, and other contaminants. The ICP-AES Spectrometer 4820/4850 features a programmable digital spark source, sensitive optical systems, and real-time drive correction, ideal for precise water quality analysis. The ICP-OES Spectrometer 6500 uses dual-view torch technology, a solid-state RF power source, and a wide-area ECCD sensor to ensure high sensitivity, even in complex matrices. Both products meet environmental compliance testing requirements and help reduce operational costs, such as argon consumption. These products, along with other AELAB spectrometers, offer a comprehensive solution for water monitoring and ensuring water safety.

Conclusion
ICP-OES is a versatile and reliable technique for monitoring water quality, especially in detecting toxic metals such as arsenic, mercury, cadmium, and lead. Its ability to analyze multiple elements simultaneously and its high sensitivity make it invaluable for regulatory testing. However, challenges related to sample matrix interferences and detection limits must be carefully managed to ensure accurate results. This technique remains one of the most widely used methods for environmental monitoring, with significant applications in drinking water quality assessment and environmental protection.