The Impact of High-Salt Matrices on ICP-MS Performance
Challenges in High-Salt Sample Analysis
When analyzing high-salt samples using ICP-MS (Inductively Coupled Plasma Mass Spectrometry), the presence of dissolved salts like NaCl, KCl, and other ionic compounds can interfere with the accuracy of elemental detection. These ions can cause spectral interferences, reduce sensitivity, and lead to poor recovery rates of target elements. Understanding the nature of these interferences is crucial for improving measurement precision.
Common Interferences in ICP-MS
High-salt matrices often lead to issues such as:
-
Isobaric Interferences
: Salts like sodium can cause interferences with the detection of elements like lithium or magnesium.
-
Matrix Effects
: High concentrations of sodium or potassium can affect the ionization efficiency, resulting in signal suppression or enhancement.
-
Cl- Complex Formation
: Chloride ions in high-salt solutions can form complexes with metals, making it harder to measure target analytes effectively.

Learn more:Vortex Mixers vs. Centrifuges: A Comprehensive Guide to Enhanced Lab Efficiency
Techniques for Overcoming Salt Matrix Interference
Use of Sample Preparation Methods
Several methods can help mitigate the effect of high-salt matrices:
-
Matrix Removal with Resin
: Special resins can be used to remove high levels of foreign ions, enriching the analyte of interest. For example, boron-specific resins can adsorb boron from high-salt matrices, allowing more accurate quantification of boron in brines and other salty samples.
-
Dilution and Filtration
: While dilution can sometimes lower the salt concentration, it often leads to lower analyte concentrations, which may fall below the detection limits. Careful filtration is also necessary to remove particulate matter.
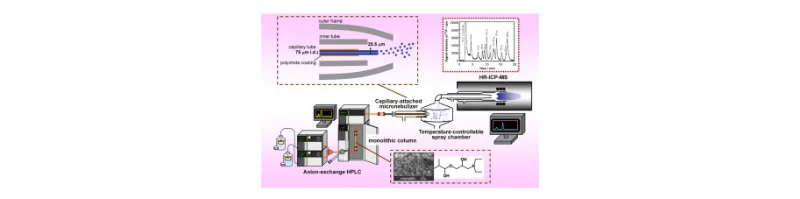
Figure 1: Integration of HPLC with ICP-MS reduces interferences and enhances accuracy by separating interfering ions before detection.
Elution and Ion Exchange Techniques
Ion-exchange resins, such as Dowex 50 W×8 and Ion Exchanger II, play a critical role in purifying high-salt samples by removing unwanted ions. These resins selectively adsorb different ions based on their charge and size. By using a combination of strong acid cation resin and weak-base anion resin, high-salt samples can be processed to achieve high recovery rates for analytes like boron, even in complex matrices.
Boron Enrichment from High-Salt Samples Using Resin Technology
Boron-Specific Resins for Accurate Detection
Boron analysis in high-salt samples, such as halite or brines, is particularly challenging due to the presence of sodium chloride and other interfering ions. However, boron-specific resins, when used in conjunction with ion-exchange techniques, can effectively remove these interferences. The boron is adsorbed as H3BO3 in the resin, and elution with HCl at elevated temperatures allows for the accurate recovery of boron from salty matrices.
ICP-OES and ICP-MS for Boron Quantification
Using the enriched sample, both ICP-OES and ICP-MS can be used to quantify boron. ICP-MS is often more sensitive, allowing detection at lower concentrations, while ICP-OES offers faster analysis times with lower sensitivity. Both techniques provide reliable results, especially when high-salt interference is removed using resin-based purification methods.
AELAB Products: Enhancing ICP-MS Performance for High-Salt Samples
The ICP-MS Spectrometer 7000 by AELAB Technology is a state-of-the-art analytical instrument specifically designed to tackle the challenges of high-salt sample analysis. With advanced features such as high-sensitivity ion interfaces, distributed collision reaction cell (CRC) technology, and robust matrix resistance for handling up to 10% high-salt matrices, it ensures accurate detection and quantification of trace elements. AELAB’s innovative solutions, including advanced ion-exchange resins, online argon dilution, and automated purification systems, provide laboratories with unparalleled performance, even in the most complex matrices. Additionally, its patented design minimizes interferences, enhances sensitivity by up to 5 times, and achieves detection limits as low as ppt, making it ideal for applications in environmental monitoring, food safety, pharmaceuticals, and semiconductor industries. With AELAB’s cutting-edge technology, laboratories can achieve optimal results in high-salt ICP-MS analysis with reliability and efficiency.

Conclusion: Optimizing ICP-MS Analysis for High-Salt Samples
The analysis of high-salt samples in ICP-MS presents numerous challenges, including spectral interferences, matrix effects, and ionization suppression. However, through the use of sample preparation techniques such as resin-based purification, dilution, and filtration, these issues can be mitigated. AELAB’s innovative products provide essential tools to overcome these challenges, enabling precise and accurate elemental analysis even in the presence of high-salt matrices. By improving sample preparation and utilizing advanced detection methods like ICP-OES and ICP-MS, laboratories can achieve reliable results in a wide range of applications.