Introduction
In molecular biology, chemistry, environmental science, and diagnostics, accurate sample analysis is essential. Two of the most widely used instruments for light-based measurements in these fields are spectrophotometers and fluorometers. While both deal with light interactions, they operate on different principles and serve distinct analytical purposes.
This comprehensive guide explores their fundamental differences, applications, advantages, and limitations to help you select the most appropriate instrument based on your experimental needs.
What is a Spectrophotometer?
Definition and Principle
A spectrophotometer is an analytical instrument that measures how much light a sample absorbs at specific wavelengths. Based on the Beer-Lambert Law, it correlates the amount of light absorbed to the concentration of analytes in the sample.
How It Works
A light source emits a wide spectrum of light.
A monochromator selects a specific wavelength.
The selected light passes through the sample.
A detector measures the amount of light transmitted or absorbed.
Applications
Measuring DNA/RNA and protein concentrations
Monitoring cell density
Environmental and water quality analysis
Chemical compound quantification
Advantages
Cost-effective and easy to operate
Broad dynamic and wavelength range
Compatible with solids, liquids, and gases
Fast measurements
Limitations
Moderate sensitivity (nanogram range)
Cannot distinguish between similar absorbing compounds
Less effective for very low-concentration samples
What is a Fluorometer?
Definition and Principle
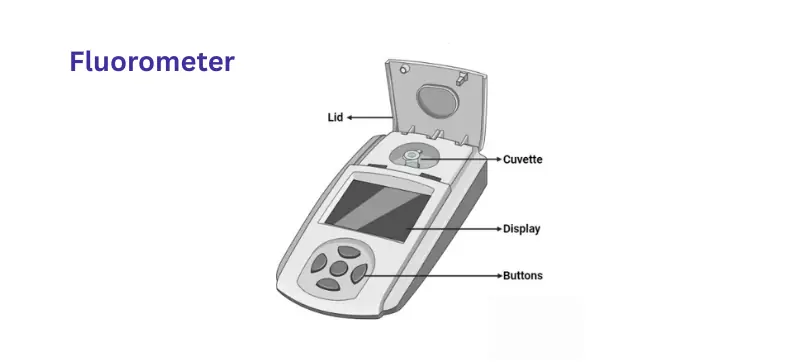
A fluorometer (or fluorimeter) measures the fluorescence emitted by a substance when excited by light. Certain molecules absorb light at one wavelength and emit it at a longer wavelength—this emission is proportional to concentration and used for ultra-sensitive detection.
How It Works
The sample is excited by light at a specific wavelength.
It emits light at a longer wavelength.
A detector captures the emitted fluorescence for quantification.
Applications
Quantification of nucleic acids and proteins
Detection of trace contaminants
Clinical assays based on fluorescence
Environmental and biochemical studies
Advantages
Extremely high sensitivity (picomolar to nanomolar)
Highly selective with specific fluorescent dyes
Ideal for low-concentration samples and molecular interactions
Limitations
Requires fluorescent dyes or naturally fluorescent molecules
Sensitive to pH, temperature, and solvent conditions
More complex setup and higher cost
Key Differences: Spectrophotometer vs Fluorometer
| Criteria | Spectrophotometer | Fluorometer |
|---|
| Measurement Principle | Measures absorbed light | Measures emitted fluorescence |
| Sensitivity | Moderate (nanogram range) | Very high (picogram range) |
| Selectivity | Low | High (with fluorescent dyes or labels) |
| Speed of Use | Fast and straightforward | Slower due to dye prep and emission detection |
| Complexity | Simple setup | Requires more careful preparation |
| Cost | Lower cost | Higher cost (instruments and reagents) |
| Best for | High-concentration, general-purpose assays | Low-concentration, highly sensitive measurements |
When Should You Use Each Instrument?
Use a Spectrophotometer When:
You are measuring moderate to high concentrations (e.g., 100–1000 ng/µL).
You require quick, cost-effective analysis.
Your samples do not need fluorescent labeling.
General absorbance-based assays are sufficient.
Use a Fluorometer When:
You need to detect very low concentrations (e.g., in the picogram range).
High sensitivity and selectivity are crucial.
Your experiment involves labeled DNA/RNA, proteins, or trace toxins.
You’re conducting advanced molecular or biochemical research.
Expert Tips for Choosing the Right Tool
Know Your Sample: Does it fluoresce naturally or require labeling?
Required Sensitivity: Choose based on the minimum detectable concentration.
Budget Constraints: Spectrophotometers are more budget-friendly.
Precision Needed: Fluorometers excel in high-precision applications.
Ease of Use vs Performance: Spectrophotometers are simpler, but fluorometers offer greater sensitivity.
Conclusion
Both spectrophotometers and fluorometers are indispensable tools in modern laboratories. Spectrophotometers offer simplicity, versatility, and affordability for routine absorbance measurements. Fluorometers, with their unmatched sensitivity and selectivity, are ideal for detecting trace amounts of molecules and conducting complex molecular analyses.
Your choice should be guided by sample characteristics, required sensitivity, analytical goals, and available resources. For general quantification and rapid assessments, a spectrophotometer is often sufficient. For precision, trace detection, or dye-based assays, a fluorometer is the clear choice.