Introduction
Taq DNA polymerase, a thermostable enzyme derived from the thermophilic bacterium Thermus aquaticus, has revolutionized the field of molecular biology. At the core of the Polymerase Chain Reaction (PCR), Taq polymerase in PCR enables the rapid amplification of DNA sequences, powering countless applications in diagnostics, research, and biotechnology.
In this article, we’ll explore what makes Taq polymerase indispensable to PCR, its unique properties, advantages, limitations, and how it continues to be a vital tool in modern science.
What Is Taq DNA Polymerase?
Taq DNA polymerase is a DNA-synthesizing enzyme known for its heat resistance and high-speed polymerization activity. It functions optimally at 70–75°C and retains activity at temperatures as high as 92°C, making it ideal for the high-temperature demands of PCR.
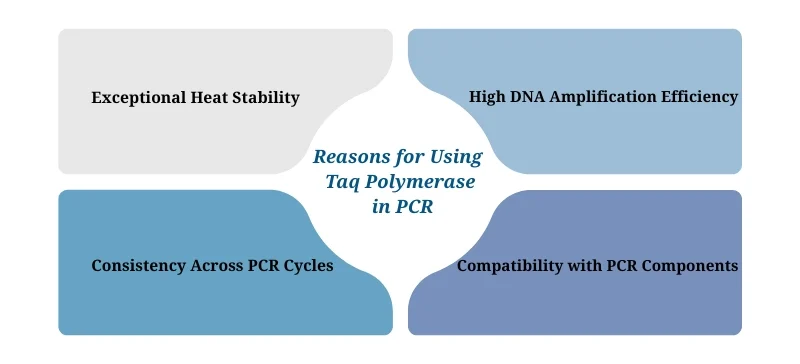
Why Is Taq Polymerase Used in PCR?
1. Exceptional Heat Stability
PCR involves repeated heating cycles that denature double-stranded DNA at ~94–98°C. Unlike typical DNA polymerases that denature at these temperatures, Taq remains functional, enabling uninterrupted DNA synthesis through multiple cycles.
2. High DNA Amplification Efficiency
Taq polymerase synthesizes DNA at a rate of approximately 150 nucleotides per second, allowing for the rapid amplification of DNA fragments up to several thousand base pairs long.
3. Consistency Across PCR Cycles
Thanks to its heat tolerance and stability, Taq performs reliably throughout the 25–40 cycles of a typical PCR run—without requiring enzyme replenishment.
4. Compatibility with PCR Components
Taq polymerase integrates seamlessly with standard PCR reagents, including primers, dNTPs, Mg²⁺ ions, and commercial buffer systems. It also tolerates additives such as DMSO and BSA.
Mechanism of Action of Taq Polymerase in PCR
Taq polymerase operates through a three-step thermal cycling process:
Denaturation (94–98°C): DNA strands separate.
Annealing (50–65°C): Primers bind to the target sequences.
Extension (72°C): Taq polymerase adds nucleotides to the primer’s 3′ end using base-pairing rules.
It catalyzes 5′→3′ DNA synthesis and forms phosphodiester bonds between nucleotides, resulting in exponential amplification of the target DNA.
Advantages and Limitations
| Feature | Advantage | Limitation |
|---|---|---|
| Thermostability | Enables high-temperature reactions | Not suitable for highly accurate applications |
| Speed | Fast amplification | No error correction |
| Broad compatibility | Works with various PCR reagents | Sensitive to temperature shifts |
| Cost-effective | Affordable and widely available | Lower fidelity than proofreading enzymes |
Compatibility with PCR Components
Taq polymerase works harmoniously with standard PCR reagents, making it highly versatile.
Compatible With:
Primers: Effectively binds at annealing temperatures.
dNTPs: Efficiently incorporates nucleotides.
Buffers: Operates in various commercial buffer systems.
Thermal Cycling: Remains active over 25–40+ cycles.
Additives: Works with DMSO, BSA, and Mg2+ enhancers.
This compatibility ensures reliable performance across a wide range of experimental conditions.
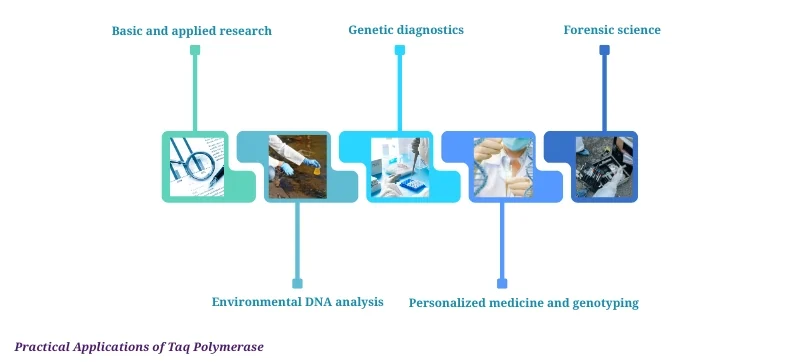
Practical Applications of Taq Polymerase
Taq polymerase powers a wide range of fields:
Basic and applied research
Genetic diagnostics (e.g., COVID-19 testing)
Forensic science
Environmental DNA analysis
Personalized medicine and genotyping
Expert Tips for Using Taq Polymerase
Use optimal Mg²⁺ concentration (typically 1.5–2.5 mM).
Store the enzyme at –20°C and avoid repeated freeze-thaw cycles.
Use hot-start formulations for complex or low-template samples.
Perform gradient PCR to optimize annealing temperature.
Conclusion: A Tried-and-True Workhorse in Molecular Biology
Despite the emergence of newer, high-fidelity polymerases, Taq Polymerase in PCR remains a cornerstone of molecular biology laboratories. Its robustness, cost-effectiveness, and ease of use make it ideal for a wide variety of PCR applications—from routine research to clinical diagnostics.